Yes, you read the title correctly. I do not have diabetes and I willingly chose to wear a CGM.
Overall, this was an eye-opening “CGM experiment” and this post covers what I learned as a non diabetic from wearing a continuous glucose monitor (aka CGM).
My CGM Experiment: What I Learned as a Non-Diabetic From Wearing a Continuous Glucose Monitor
I wasn’t sure I’d write much about it, but when I asked folks on my email list if they’d be interested in hearing about this CGM experiment (which I lovingly referred to as my semi-bionic arm), the response was HUGE.
I even polled my Instagram followers in my stories and 99% said “yes, tell me more about what you learned from wearing a continuous glucose monitor.” (Not sure why 1% aren’t interested, but I’m gonna go with majority vote here!)
I also received A LOT of specific questions about this:
- Why did you choose to wear a CGM?
- Which CGM did you choose?
- What was your average blood sugar?
- How did you get a CGM?
- How much does a CGM cost?
- How bad does it hurt to put in the CGM sensor?
And on and on and on. I received over 60 questions, so I obviously can’t answer them all here, but I’ll at least cover the biggest and most common questions.
Per usual, what I had envisioned as a short ‘n’ sweet write up turned into a bit of a research beast. I should know myself by now, ha!
Why did I wear a continuous glucose monitor (CGM)?
There are several reasons.
1. I’ve been curious about my blood sugar patterns ever since my interesting experience with taking the glucola (glucose tolerance test) during my pregnancy several years ago to screen for gestational diabetes—and my results from home blood sugar monitoring during the few weeks following that test. In short, I wanted to check on my non-pregnant blood sugar balance beyond standard labs.
2. Second, several mamas in my Real Food for Gestational Diabetes Course have used CGMs to better understand and manage their blood sugar levels. In one case, CGM readings revealed really interesting blood sugar patterns overnight and at fasting (first thing in the morning) that ultimately prevented misdosing medications that could have caused hypoglycemia overnight.
In another case, CGM data made a participant realize her blood sugar was spiking sooner and higher than she had anticipated after certain meals. With solely relying on fingersticks to check her blood sugar, she had been missing the “highest highs.” She was able to pinpoint what was causing her blood sugar spikes and better tailor her diet/lifestyle (which ultimately helped her stay diet controlled and have the out-of-hospital birth she had hoped for).
In other words, using a CGM was like having a window into their blood sugar patterns. It eased stress, improved their sense of control over what otherwise feels like a rollercoaster of a ride, and helped them better manage their gestational diabetes overall. As a certified diabetes educator, I’ve found CGMs cut out a huge amount of guesswork for clients and care providers alike.
3. Third, I had comprehensive blood work done a few months ago, which included a variety of metabolic markers. My HOMA-IR, a marker of insulin resistance, was almost too good (indicating favorable insulin sensitivity) and my fasting insulin and blood sugar were relatively low. While my results were overall very good, I was curious about what was going on with my blood sugar on a day-to-day basis.
4. Fourth, I have a family history of type 2 diabetes and reactive hypoglycemia. I know from all of my mindful eating observations that I feel best eating a moderately low carb diet, which probably is more kind to my pancreas (thanks to avoiding major blood sugar spikes). I know that heredity plays a role in your risk for disease, but also that lifestyle is a powerful way to moderate that risk. If we can catch any blood sugar issues very early on, we can take action to help prevent the progression to type 2 diabetes. If I inherited a crappy pancreas (no offense, pancreas… I don’t know for sure; I still love you man!), maybe I should take extra precautions?
If I can sum it up in one word CURIOSITY is what made me do this CGM experiment.
How do continuous glucose monitors work?
CGMs use a “minimally invasive electrochemical sensor” that’s inserted below the skin to measure blood sugar levels in interstitial fluid (that’s the fluid between your cells).
It emits a low frequency signal to communicate blood sugar data to a reader device. Different CGM systems vary in the specifics, but you essentially end up with a vast amount of data on your blood sugar patterns. Instead of relying on fingersticks and a regular glucometer, you can keep tabs on what’s happening 24/7 (without having to constantly prick yourself, hallelujah!).
How do you choose which CGM to get?
First off, this is not a sponsored post whatsoever. I had to pay out of pocket for everything (see below). I WISH the manufacturer would have donated one of these things to me since I’m about to give it a ton of free PR. The things I do for science!
I chose the Freestyle Libre for ease, functionality, and cost. Most CGMs require calibration with fingersticks using a glucometer (often with twice daily). Freestyle Libre is factory calibrated, so no need to poke your fingers all the time. This is both a benefit and a limitation of the system. It also tends to be less expensive than other CGMs.
It’s way beyond the scope of this post to discuss pros and cons of all CGM systems, so please take up that conversation with your care provider if you’re interested.
How do I get a CGM if I don’t have diabetes?
Continuous glucose monitors are by prescription only. Yes, they are typically used for people with diabetes. Even 10 years ago, it was pretty rare for anyone other than those with type 1 diabetes to get one, but new technology (that’s more accurate and affordable), provider awareness, and consumer demand has driven many non-diabetics to try out CGMs for themselves.
I simply talked it over with my doctor and he was happy to write a prescription for one. I have a feeling that my professional work as a dietitian and certified diabetes educator specializing in gestational diabetes (as well has our talk about some research studies on GD I’m consulting on) helped sway the decision in my favor.
If in doubt, it doesn’t hurt to ask! If your provider errs on the side of preventative medicine or functional medicine, I’d wager they’d be more likely to recommend a CGM.
UPDATE: One other option for obtaining a CGM is to use a company like Levels, which will connect you with a provider to see if you qualify for a CGM. This was not an option at the original publishing of this article, hence the update here. Use this link to get to the top of the 100k+ waiting list.
How much does the Freestyle Libre cost?
This will vary by your location, your pharmacy, and whether insurance covers it or not. For me, insurance did not cover it. The only pharmacy near me that carried the Freestyle Libre was Walmart, which apparently has great pricing on prescriptions.
The Freestyle Libre has two components to function: a reader and a sensor.
Reader: $79.45 (you only buy this once)
Sensor: $43.66 (you have to replace this every 10-14 days)*
Another colleague who has one (but lives in a different part of the U.S.) paid $110 total for the reader and her first sensor; replacement sensors were ~$40. As I said, this will vary based on where you live.
The cool thing is the reader doubles as a regular glucometer, so if you don’t have a blood sugar meter already, you can use this for double duty. It uses the Freestyle Precision Neo test strips.
*At the time of writing the 10-day sensor is the current norm in the U.S. They also manufacture a 14-day reader that was recently approved by the FDA (which, for reasons beyond me, is already the standard in other countries).
UPDATE: The 14-day sensor is now available in the United States. A reader device is not necessarily needed as most smart phones can serve as a reader with a special app.
Did it hurt to insert the Freestyle Libre sensor?
I’ll admit, the insertion of the sensor was something I feared most. The thought of having a sensor under my skin freaked me out, then thinking about how I would get it there freaked me out more.
Luckily, they’ve designed the Libre to be pretty foolproof. The sensor comes with a contraption that takes all the guesswork out of insertion. It’s essentially a plastic device with a spring-loaded needle—ok, that sounds scary when I write it out…. It’s a plastic thing that attaches to the sensor. You line it up on the right area of your arm and push down. Before you can blink, the spring-loaded needle thing has poked a hole, retracted, and in its place left the sensor attached to your arm.
I swear it was just as easy as a fingerprick. It did not hurt at the moment, but it does make a tiny wound where the sensor is implanted under your skin. For me, that was a little bit sore, like a very very very dull ache (pain scale 1/10) the next day, akin to the feeling after getting blood drawn.
Short answer: no, it doesn’t hurt.
Was it uncomfortable to wear a CGM?
Not really. For the Freestyle Libre, the sensor is worn on the back of the upper arm (tricep area). The part of the sensor that’s external is a little larger than the size of a quarter. The internal sensor is ~ a quarter inch long and aside from the very mild ache I had the day after placement, it didn’t bother me.
I was worried the adhesive on the sensor would irritate my skin, as I’m very sensitive to the adhesive on bandages/tape, but this was surprisingly a non-issue for me.
After a few days, I became less aware of something being stuck on my arm. I’d accidentally bump it every once in a while or catch it on a shirt as I was getting dressed or notice it as I rolled over in bed, but it otherwise it wasn’t bothersome.
I will say, I’m extremely grateful that I don’t have to wear one at all times. I can only imagine what my friends/clients with type 1 diabetes have to put up with having a CGM and often an insulin pump constantly attached to their body. Just one more thing to always have on the back of your mind.
Was the CGM easy to use?
This shows how easy it is to take a real time blood sugar reading from the Freestyle Libre continuous glucose monitor. Press the button on the reader, bring it close to the sensor on your arm, and voila!
What are normal and abnormal blood sugar levels?
This is a surprisingly hard question to answer. If you go by American Diabetes Association standards, “normal” blood sugar is less than 100 mg/dl fasting (such as first thing in the morning before eating), and less than 140 mg/dl two hours after eating.
In general, they state that non-diabetic people have blood sugar in the range of 70-130 mg/dl.
For an official diagnosis of type 2 diabetes, fasting blood sugar would be 126 mg/dl or higher, and 200 mg/dl or higher 2 hours after a 75 gram glucose tolerance test (and/or A1c of 6.4% or greater).
For an official diagnosis of prediabetes, blood sugar levels are below the criteria for type 2 diabetes, but above 100 mg/dl fasting and 140 mg/dl 2 hours after a glucose tolerance test (and/or A1c of 5.7% of greater).
What is not specified in these guidelines is how high it’s “normal” to see your blood sugar spike after meals. Since these guidelines are focused on diagnosing and treating diabetes/prediabetes, they don’t give us much insight into truly normal blood sugar levels. I’ll explain more on this later in this post.
Lily, what did you learn about your blood sugar from wearing a CGM?
This is obviously the reason you’re here. This is the reason I did the whole CGM experiment in the first place.
In some ways, my results were surprising and in other ways, exactly as I would expect. My goal was to test my regular diet and see how my blood sugar fared with my usual low-ish carb, real food, mindful eating approach. There were a few unusual meals, including Thanksgiving dinner and the morning I intentionally ate oatmeal (more on that later!); otherwise, I was just eating as I always do.
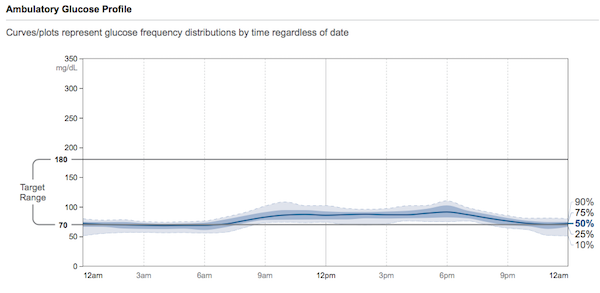
My average blood sugar over the 10 days was on the low end: 79 mg/dl. No, I did not have symptomatic hypoglycemia. The lower average was due to my blood sugar running primarily in the 60’s and 70’s at night (“fasting”). During the day, my blood sugar mostly hung out in the 80’s-90’s with occasional peaks depending on the carbohydrate content of my meals.
My lower carb real food meals rarely spiked me above 100-110. I had a few higher spikes from meals that included a small to moderate amount of carbs, such as: a slice of real sourdough, rice, potatoes/sweet potato, or hidden sugar (psst – scroll down for my oatmeal experiment).
For example, we had some Thai food from a local Thai restaurant and I’m pretty sure the curry and sauces have sugar in them (it’s pretty standard in Thai cooking to add sugar). I was surprised after eating a big bowl of curry with chicken and vegetables and the tiniest serving of white rice (we’re talking ⅓ cup, friends), to see my blood sugar spike to the 130’s.
Even though the rice was leftover (therefore had been cooled for >12 hours, which converts a portion of the starch into “resistant starch”, which is praised as healthful for our microbiome and less-impactful on blood sugar levels), it clearly still spiked my blood sugar. I can personally feel my blood sugar spiking at fairly low levels with symptoms like racing heart and jitteriness. It’s subtle, but noticeable, probably because my blood sugar tends to run stable in the lower range.
I repeated the same Thai food leftover meal the next day, just without the rice (so I’m sure there was still hidden sugar in the curry) and my highest peak was only 105. White rice and I are apparently not friends, even in portions that provide only 20 grams of carbs.
Sorry, everyone who’s #teamwhiterice. It doesn’t work for me.
Does my blood sugar run low, are the criteria for “normal blood sugar” wrong, or was my sensor off?
I’ll be honest, seeing my numbers average on the low end made me question a lot of things.
First off, is my blood sugar truly low? Maybe. Last time I had blood work drawn (meaning a venous blood sample), my fasting glucose was 68 mg/dl. This was after a 14 hour fast (not intentional; my appointment got pushed back that day).
This makes me think the Freestyle Libre readings were accurate. My fasting insulin levels were also on the low side, which is physiologically to be expected when fasting. The body is going to preferentially switch to burning fat/ketones and meanwhile both glucose and insulin will decrease during extended periods without food.
I spot tested with my glucometer a few times to check the accuracy of the CGM. It was always within 10 mg/dl of my fingersticks, but interestingly the “lower” values from the CGM tested 5-10 points higher on fingersticks, and the “higher” values on the CGM tested 5-10 points lower when double checked with the fingersticks (in other words, CGM made my lows look lower and my highs look higher than was indicated by my blood sugar meter).
My readings were still within an acceptable range of variation, but I did dig up a few articles in the medical literature that suggest that Freestyle Libre readings can be a little off, either from a factory calibration error or other factors. Even so, observing the overall patterns in my blood sugar was invaluable.
It’s also well-known that interstitial glucose (what you measure with a CGM) can have a lag time compared to capillary glucose (what you measure with a finger stick). That in and of itself could explain the slight discrepancy I observed between fingersticks and CGM readings.
If I assume that my blood sugar truly runs on the lower end of the normal range, maybe our current accepted “norms” for blood sugar aren’t perfect.
As with most lab values, the normal range is defined by a certain number of standard deviations from the average. I may be a person who naturally runs on the lower end of the range.
Nonetheless, the research thus far suggests that:
“In individuals with normal glucose tolerance, glycemia is maintained within a narrow range between 68.4 and 138.6 mg/dL.” (Diabetes & Metabolism Journal, 2015)
I tried to dig up some data on blood sugar patterns in hunter-gatherers (modern-day, obviously), to shed some light on the issue.
In the Hadza (people who are indigenous to Tanzania), researchers have yet to observe fasting glucose readings above 85 mg/dl. Similarly, fasting glucose levels from the Shuar (indigenous people of Ecuador and Peru), run on the lower side, with “fasted glucose levels among rural Shuar men (73.6 ± 13.2, n = 32) and women (82.1 ± 21.2 mg dL−1, n = 49).” (Obesity Reviews, 2018)
That means some indigenous people subsisting on their traditional diet have fasting glucose levels as low as 60 mg/dl.
Perhaps my blood sugar readings in the 60s and 70s at night aren’t so low after all?
Are you sure your blood sugar is running low? Enter: OATMEAL.
Another interesting finding was my oatmeal test. Now, for the duration of the experiment, I ate my normal breakfast, which is typically some variation of 2 eggs cooked in butter or lard, vegetables (usually rotating between kale, broccoli, mushrooms, onions, etc.), and possibly some breakfast meat, like sausage or bacon. Alongside this, I have black tea (unsweetened) with heavy cream. [Read: I eat a low carb, high fat, moderate protein breakfast.]
This style of breakfast was a dream for my blood sugar, essentially flatlining it in the 80s or 90s. It’s also excellent for my energy levels, satiety, and productivity (low carb + real food + mindful eating for the win!). I can easily go for 3-5 hours without getting hungry (depends on the day and how active I am), which is nothing short of a miracle for someone who used to be a huge snacker.
There’s a reason I tend to return to a variation of this day after day. Even when I added a small slice of sourdough one morning and ½ cup of leftover roasted potatoes another morning, my blood sugar didn’t exceed 100 (the magic of not eating “naked carbs”).
I started to wonder if I was just super insulin sensitive in the morning or maybe had more wiggle room for carbs.
So, the final day of my CGM sensor, I decided to eat a breakfast similar to the Academy of Nutrition and Dietetics’ sample meal plan in their pregnancy guidelines. Their meal plan is oatmeal, skim milk, and strawberries. In other words, all carbs.
(For those of you familiar with my book, Real Food for Pregnancy, this is the same meal plan that I use to make a comparison on the nutrient-density and macronutrient ratio compared to my real food meal plan. This excerpt of the book is included in the free chapter download; see the bottom of this post or this page to get it.)
I don’t have skim milk in the house (and I never will), but I do have whole, grass-fed milk for my son. I also didn’t have strawberries, but I have raspberries, which are nutritionally similar. So I whipped up measured portions of rolled oats (1/2 cup dry, which is 1 cup cooked) prepared with water, a little milk poured on afterwards (I’m really not a fan of straight up milk, so I only used a few Tbsp), ½ cup raspberries, and because it was in-edibly plain, I added 1 measured teaspoon of honey (not heaping). Total carb count was 45g.
All things considered, this was NOT a large bowl of oatmeal and it was essentially NOT sweet, despite adding a little honey. (I say this to point out that the average person adds A LOT of extra sweeteners to their oatmeal, either with sugar/honey or dried fruit. My version would be unpalatable to many people.)
At first, I thought my blood sugar was doing ok after the oatmeal, but I then watched with horror on my Freestyle Libre as the readings climbed. When you scan the sensor, the Freestyle Libre reader shows an arrow next to the numerical reading with an up, down, level, or slightly up/down error, indicating your real time blood sugar trends. This was the ONLY time during the entire 10 days that I saw the straight up arrow, indicating my blood sugar was rising FAST.
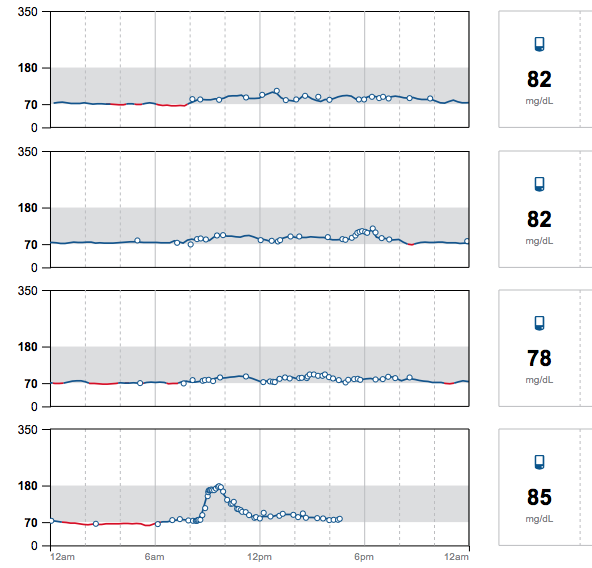
My blood sugar went from 74 to a peak of 178 in an hour. By two hours, I was down to the 120s and by three hours, finally back down to 100.
This perfectly illustrates why I preach “no naked carbs” (meaning carbs eaten without a source of fat or protein). The spike from oatmeal was 40 points higher than my Thanksgiving dinner, which included stuffing, sweet potatoes, cranberry sauce, pumpkin pie (in other words, the same or more carbohydrates than this unsatisfying, bland oatmeal breakfast). I’d wager that I didn’t spike as high from Thanksgiving because there was sufficient fat, protein, and fiber in the meal to slow digestion and absorption of the carbohydrates.
This image shows 4 days of blood sugar data. The first 3 days, I’m eating my regular diet. The last day, I did the oatmeal experiment. See the peak on day 4?
What was interesting, though, is that I got ravenously hungry when my blood sugar started plummeting. Now, this is a mindful eating/blood sugar physiology teaching I’ve been using in practice for years (go back to my viral post, The Healthy Breakfast Mistake, for a hilarious run down).
It was really interesting to watch it pan out in real time on my CGM. I had always assumed that I wouldn’t be hungry until my blood sugar was back down to normal or in the hypoglycemic range, but this experiment showed me that the hunger trigger (for me, anyways) is in response to the impending crash.
I actually didn’t end up hypoglycemic in response to this meal, but I literally had to eat something with substance (fat/protein) around the two hour mark (~ 2 oz of leftover grass-fed beef burger patty) to avoid going hangry. That probably stopped me from going into reactive hypoglycemia.
After this oatmeal experiment, I started wondering if my glycemic response to oatmeal was exaggerated or unusual. It’s surprisingly hard to find data on blood sugar responses in non-diabetics, especially when trying to examine the “peak” blood sugar response.
As you can recall, as long as blood sugar is back down to 140 mg/dl by two hours after eating, then you’re supposedly “in the clear” by conventional guidelines for diabetes/prediabetes. If a study has people measure their blood sugar only at 2 hours, you’re likely to miss the peak glycemic response in many people. Moreover, different people peak at different times, so without a million finger pricks or CGM, the results aren’t going to be very meaningful.
Blood sugar patterns in non-diabetics
After spending entirely too much time on Google Scholar (this is my M.O. in life), I was able to find some studies on continuous glucose monitoring in non-diabetics (hooray!).
In one study from Stanford, 57 participants—comprised of people with and without diabetes—wore a CGM for 2-4 weeks. (PLoS Biology, 2018)
They stratified the glucose data into 3 subtypes of glycemic variability (essentially, how much a person’s blood sugar spiked or dropped throughout the day). The healthiest subjects tended to have the lowest glycemic variability. In other words, their blood sugar stayed within a tightly controlled range for most of the day.
These people tended to have a lower BMI, be younger, and have lower readings of glucose, A1c, fasting insulin, and triglycerides (this resembled my glycemic patterns).
A subset of the study group were given standardized meals to measure the glycemic response. In the test of cornflakes and milk (which is nutritionally similar to my oatmeal breakfast), fully 80% of people without diabetes experienced blood sugar spikes beyond 140 mg/dl (prediabetic levels). Furthermore, 23% of this group saw blood sugar levels exceeding 200 mg/dl (diabetic levels) after cornflakes and milk.
They write:
“It is interesting to note that although individuals respond differently to different foods, there are some foods that result in elevated glucose in the majority of adults. A standardized meal of cornflakes and milk caused glucose elevation in the prediabetic range (>140 mg/dl) in 80% of individuals in our study. It is plausible that these commonly eaten foods might be adverse for the health of the majority of adults in the world population.” (PLoS Biology, 2018)
In a press release on the study, one of the authors comments further on the cornflakes result:
“Make of that what you will, but my own personal belief is it’s probably not such a great thing for everyone to be eating.”
Ya think?
This is the whole argument for a lower carbohydrate diet as a treatment and/or preventative strategy for type 2 diabetes.
They also note that occasional high blood sugar readings (arguably, not ideal), are common, even among people without diabetes:
“Importantly, we found that even individuals considered normoglycemic by standard measures exhibit high glucose variability using CGM, with glucose levels reaching prediabetic and diabetic ranges 15% and 2% of the time, respectively. We thus show that glucose dysregulation, as characterized by CGM, is more prevalent and heterogeneous than previously thought and can affect individuals considered normoglycemic by standard measures, and specific patterns of glycemic responses reflect variable underlying physiology. The interindividual variability in glycemic responses to standardized meals also highlights the personal nature of glucose regulation.” (PLoS Biology, 2018)
This study brings into question the overall insistence on using glucose tolerance tests, A1c, and fasting blood sugar to define diabetes, as if it’s this black and white diagnosis. (Spoiler alert: it’s not.)
Imagine if you could go to the doctor, have a CGM sensor inserted on the spot, then return in 2 weeks to have your data analyzed and graphed using the criteria in this study. It would give us a much more nuanced look into your blood sugar regulation. It would also reveal what foods are great for your blood sugar and conversely, which ones are a glycemic disaster. This would be preventative medicine. Intervene before blood sugar levels are consistently in the diabetic range, before insulin resistance gets too severe, and before beta cell burnout.
Then again, this would be a royal pain for practitioners/insurance companies who like the black and white nature of single tests. I don’t think I’ll hold my breath waiting for this to become the standard of care.
In another CGM study (this one from Spain), glycemic patterns were tracked in 322 people (both prediabetic and non-diabetic) eating their usual diets, providing 1521 complete days of data. (Diabetes Research and Clinical Practice, 2018)
Participants had blood sugar in the non-diabetic range (<140 mg/dl) 97% of the time.
Interestingly though, 73% of participants experienced episodes of high blood sugar in the prediabetic range (and 5% in the diabetic range).
The average amount of time spent above the normoglycemic range (blood sugar over 140 mg/dl) was 32 min per day in the normoglycemic group and 53 min in the prediabetic group. The peak blood sugar reading for non-diabetics averaged 119 mg/dl (compared to 128 mg/dl in the prediabetic group).
Also interesting was that 44% of non-diabetic people in this study had some bouts of “hypoglycemia” defined as less than 70 mg/dl. (This again made me question if 70 mg/dl is really an appropriate cut off for hypoglycemia.)
I think this data highlights just how hard our bodies work to keep our blood sugar in a tightly controlled range 24/7. I don’t think a few high readings are necessarily a sign of something wrong, it just shows you which foods force your body to work especially hard to bring those “highs” back to the normal range as quickly as possible.
Blood sugar patterns in non-diabetic people who are morbidly obese
Another interesting dataset I came across was an analysis of CGM data from morbidly obese participants with and without prediabetes (literally these participants were applicants to The Biggest Loser; it’s in the study methods) found significantly higher glycemic variability and overall higher glucose levels when compared to CGM data from non-diabetic, non-overweight adults. (J Diabetes Sci & Tech, 2014)
Specifically, prediabetic Biggest Loser applicants had 17.6% of blood sugar readings above 140 mg/dl while non-diabetic Biggest Loser applicants had 11.8%. Meanwhile, non-diabetic adults who are not morbidly obese only experienced 0.3-4.1% of readings above 140 mg/dl.
This data highlights the role that excess body weight plays in blood sugar regulation, even if you “pass” diabetic/prediabetic screenings by conventional standards.
Should we be eating so many carbs?
All things considered, I think what these studies highlight is that modern, high-carb diets are a mismatch for our physiology. The research has a name of it: evolutionary discordance hypothesis.
In other words “departures from the nutrition and activity patterns of our hunter‐gatherer ancestors have contributed greatly and in specifically definable ways to the endemic chronic diseases of modern civilization” (Nutrition in Clinical Practice, 2010)
To quote Eric Sodicoff, MD, “CGM is useful to prove that you need to be eating stone age food.”
Is there harm—or even benefit—in occasional blood sugar excursions?
Some argue that elevated blood sugar after meals is normal, while others argue that you should aim to keep your blood sugar as “flatlined” as possible. Honestly, I don’t know which is true and I don’t think we have an easy answer.
Where we need more research is in blood sugar patterns of metabolically healthy individuals. I’d be especially curious to see some CGM data from modern day hunter-gatherers.
We have an abundance of data on the harms of consistently elevated blood sugar (and certainly as a country are observing first-hand epidemic levels of beta cell burnout and insulin resistance). On the other hand, I can see from the perspective of maintaining metabolic flexibility (“testing” your pancreas just like you “test” your immune system during occasional illness, seasonal differences in food availability ancestrally) that it may be harmless to experience occasional blood sugar spikes.
From an ancestral perspective, carbohydrate intake varied seasonally. So It’s certainly plausible that having some occasional high blood sugar readings is not a concern, so long as there are spans of time where they can return to baseline (which was likely the case before the convenience of a globalized food supply, grocery stores, and take out).
The key here is that if occasional blood sugar spikes are ancestrally normal, it was also ancestrally normal to have periods of time WITHOUT blood sugar spikes. That is something that is missing from modern life for most of us.
Moreover, as I cover in Real Food for Pregnancy, the most comprehensive assessment of dietary patterns in modern-living hunter gatherers shows that they eat far fewer carbohydrates than we do with an average of 16-22% of calories from carbs. Compare that to our dietary guidelines, which push 45-65% of calories from carbs. It’s just A LOT of carbs for the human body to process.
Studies looking at glycemic variability, which refers to swings in blood glucose levels, tend to point towards minimizing the frequency of blood sugar spikes for heart health:
“Since 1997, more than 15 observational studies have been published showing that elevated [postprandial glucose], even in the high nondiabetic [impaired glucose tolerance] range, contributes to an approximately 3-fold increase in the risk of developing coronary heart disease or a cardiovascular event [13]. Moreover, the meta-analysis of the published data from 20 studies of 95,783 individuals found a progressive relationship between the glycemic variability (GV) and cardiovascular risk [49]. In summary, the accumulated data that GV seems to be associated with the development of microvascular complications appear to be impressive.” (Diabetes & Metabolism Journal , 2015)
Bigger implications for diabetes and prediabetes
At this time, 49-52% of the adult US population has either diabetes or prediabetes, many of whom remain undiagnosed. (JAMA, 2015) It’s estimated that upwards of 70% of people with prediabetes will go on to develop type 2 diabetes. (PLoS Biology, 2018)
A 2018 analysis of the most recent National Health and Nutrition Examination Survey data found that only 12% of Americans are “metabolically healthy.” TWELVE PERCENT. (Metabolic Syndrome & Related Disorders, 2018).
That means 88% of Americans are NOT metabolically healthy.
By the way, their definition of metabolic health included fasting blood sugar, A1c, blood pressure, triglycerides, HDL cholesterol (checking to see they had sufficient “good” cholesterol), and waist circumference. I was pleased to see they left total and LDL cholesterol as well as BMI out of their criteria, as these are less reliable markers of overall health.
I have no doubt that if I continued to eat high-carb meals for my lifetime (like oatmeal), pushing my blood sugar levels to prediabetic levels again and again and again… gradually resulting in a breakdown of my insulin sensitivity, I would become one of these statistics. Furthermore, eating this way leaves me feeling like garbage. It makes me crazy hungry for all the wrong foods as my poor body attempts to regulate blood sugar between spike after spike.
Fortunately, I don’t have to go down that path because this experiment has highlighted just how crucial a lower-carb, real food diet is to maintaining blood sugar balance, even for people like myself, who supposedly have normal glucose tolerance. If you watch the spectacular results of the Virta program or similar diabetes interventions using a lower carbohydrate approach, it’s just common sense. (Diabetes Therapy, 2018; JMIR Diabetes, 2018)
Even without blood sugar data, simply relying on mindful eating is what keeps me eating this way. If I feel good, if my energy is solid, if my mind is clear, if I’m sleeping well, if my menstrual cycles are normal, if my digestion is happy, if my thyroid is normal… obviously my body is communicating that this works for me. I can check all of those boxes (well, now that my toddler sleeps, I can check the sleep box… hooray!).
Furthermore, all my other lab markers of blood sugar and insulin regulation are good. Fasting insulin is low, HOMA-IR (a marker of insulin resistance) is good, leptin:adiponectin ratio is good (this is another surrogate marker of insulin resistance), A1c is good, all lipids are optimal, CRP is low, BMI is normal, etc.
The point is that even if you’re “metabolically healthy,” your blood sugar can still spike pretty high if you eat a bunch of naked carbs.
My oatmeal experiment made that crystal clear.
Why is carb tolerance so individual?
Everyone’s carb tolerance is different and that’s what is so cool about wearing a continuous glucose monitor. For me personally, total carbohydrate intake at meals was the biggest determining factor of my blood sugar response. Non-starchy vegetables, which contain small amounts of carbohydrates but a higher proportion of fiber (meaning low in net carbs), didn’t spike my blood sugar whatsoever, even at meals with BIG portions of non-starchy vegetables.
I was able to tolerate potatoes, sweet potatoes, winter squash, beans, real sourdough bread, and fruit (eaten whole) without a spike my blood sugar so long as I ate them in reasonable portions (about half a cup at a time) and alongside fat and protein (thus lowering the glycemic load) and lots of non-starchy vegetables (fiber & phytochemicals). I learned that rice, oatmeal, and hidden sugar did not work well for my body.
It could be that the latter sources of carbohydrates are “acellular,” which some research argues has a more severe glycemic and inflammatory response.
It could be that my microbiome has some preferences of its own (yes, the bacteria in your gut appear to play a role in your blood sugar).
It could be that I inherited a pancreas that doesn’t enjoy pumping out big boluses of insulin (could be from generations past or even my mom’s diet while I was in utero). Yay, #epigenetics.
Technically, it doesn’t matter what the mechanism is. What matters is that I can identify the foods that spike my blood sugar and make a choice as to if I eat them, in what portions, and how often.
Final thoughts on my CGM experiment
There’s a lot of “noise” in the nutrition world.
People telling you that you need to eat a certain macronutrient ratio. People telling you that going low carb is guaranteed a bad choice for women (there’s some nuance here, but I overall disagree so long as you’re eating sufficient quantities of nutrient-dense food, managing your stress, and not over-exercising). People telling you that you must go plant-based or carnivore.
Essentially we have a lot of people telling you that whatever you’re doing is wrong.
Wearing a CGM cuts through all the noise.
Eating for better blood sugar balance has carryover benefits to the rest of your health. I think the key with CGM is to try to observe other symptoms and how they relate to your glycemic response to a meal.
Note how your energy and hunger/fullness levels relate to your blood sugar.
Note how your sleeping patterns affect your blood sugar.
Note how eating to satiety vs. eating to discomfort affects your blood sugar.
Note how snacking or not snacking affects your blood sugar.
Note how eating an earlier or later dinner affects your blood sugar.
Note how exercise, stress, meditation, deep breathing, light exposure, nutritional supplements, etc. all affect your blood sugar.
Then play around with all of these factors to make adjustments to bring your blood sugar back to a healthy range.
I see CGM as a huge opportunity for preventative medicine. Here’s to hoping this technology becomes available more widely, for a lower cost, and without a prescription.
If you have questions or comments on CGM, please share them below and I’ll do my best to address them here or in a future blog post/interview.
Until next week,
Lily
PS – I know a big portion of my audience is interested in pregnancy. If you’re curious how blood sugar balance is affected by pregnancy, I encourage you to check out Ch 9 of Real Food for Pregnancy, where I sort through all the pros/cons of the different testing options for gestational diabetes and how blood sugar patterns naturally shift during pregnancy.
Also, see this post that explores 9 myths about gestational diabetes.
Research into using CGM as a replacement for glucose tolerance tests are underway, so we’ll hopefully have more insight into this as a screening option for gestational diabetes sometime in the future.
References
1. Suh, Sunghwan, and Jae Hyeon Kim. “Glycemic variability: how do we measure it and why is it important?.” Diabetes & metabolism journal 39.4 (2015): 273-282.
2. Pontzer, H., B. M. Wood, and D. A. Raichlen. “Hunter‐gatherers as models in public health.” Obesity Reviews 19 (2018): 24-35.
3. Hall, Heather, et al. “Glucotypes reveal new patterns of glucose dysregulation.” PLoS biology 16.7 (2018): e2005143.
4. Rodriguez-Segade, Santiago, et al. “Continuous glucose monitoring is more sensitive than HbA1c and fasting glucose in detecting dysglycaemia in a Spanish population without diabetes.” Diabetes research and clinical practice 142 (2018): 100-109.
5. Salkind, Sara J., et al. “Glycemic variability in nondiabetic morbidly obese persons: results of an observational study and review of the literature.” Journal of diabetes science and technology 8.5 (2014): 1042-1047.
6. Konner, Melvin, and S. Boyd Eaton. “Paleolithic nutrition: twenty‐five years later.” Nutrition in Clinical Practice 25.6 (2010): 594-602.
7. Menke, Andy, et al. “Prevalence of and trends in diabetes among adults in the United States, 1988-2012.” Jama 314.10 (2015): 1021-1029.
8. Hall, Heather, et al. “Glucotypes reveal new patterns of glucose dysregulation.” PLoS biology 16.7 (2018): e2005143.
9. Araújo, Joana, Jianwen Cai, and June Stevens. “Prevalence of Optimal Metabolic Health in American Adults: National Health and Nutrition Examination Survey 2009–2016.” Metabolic syndrome and related disorders (2018).
10. Saslow, Laura R., et al. “Outcomes of a Digitally Delivered Low-Carbohydrate Type 2 Diabetes Self-Management Program: 1-Year Results of a Single-Arm Longitudinal Study.” JMIR diabetes 3.3 (2018): e12.
11. Hallberg, Sarah J., et al. “Effectiveness and safety of a novel care model for the management of type 2 diabetes at 1 year: an open-label, non-randomized, controlled study.” Diabetes Therapy 9.2 (2018): 583-612.









Thank you for this excellent article Lily. Really interesting!
Happy New Year, and keep up the great work!
Thank you! Glad you enjoyed it Afifah.
Great piece. I had *exactly* the same experience. Non-diabetic, low-carber, wore a Libre for a month and tasted a wide range of foods. No scrip needed at the time (a year ago) in Canada.
No script? That would be amazing to see in the states!
Check out NutriSense.io
We issue prescriptions for non-diabetics, deliver CGMs right to your door, and our app is specific for non-diabetics looking to track and optimize health.
Wow, that was very insightful!! Thank you for doing the legwork to get this info. I read your book after being diagnosed with GD, and followed that (as well as a Trim Healthy Mama S meal approach) for the remainder of my pregnancy. I’m still doing random glucose checks (fasting and the odd post-prandial) to see how my body responds to carbs and sugar (we eat a mainly keto diet, with the odd splurge), and I find a CGM so fascinating. I might see if my doctor will prescribe one even though I’m not diabetic. Thank you for sharing this wealth of knowledge!
Glad to hear Real Food for Gestational Diabetes was helpful to you! I hope you can get your hands on a CGM for your own experiment.
That was a fascinating write up! You are by far the standout in this space regarding pregnancy nutrition and healthy diet. So scientifically sound, honest, reasonable and real. Thanks for this detailed article!
Glad you enjoyed it!
Thanks so much for doing this experiment and writing it up. I can’t say I’m surprised by any of it as it confirms what we both expected, but it’s helpful to have this information out there for people to gain a better understanding of how “healthy” foods are really just contributing to metabolic dysfunction.
I’m also not on #teamwhiterice despite being half-Persian. If I eat any more than a 1/2 cup of rice when eating out, my husband needs to drive us home because I fall asleep on the way home
It certainly confirmed a lot of my mindful eating observations!
This is super interesting!! I want to monitor mine as well. I still finger prick often even after a great A1C. I am just so curious and cautious…and I find it so fascinating!
The whole naked carb thing fascinates me too. I find if my son eats any sort of naked carbs he goes batshit crazy. It’s really interesting. But paired with fats/protein he has a complete different reaction. Not diabetes related but because of you I tried experimenting with this and it seriously makes a big difference in his behaviour and mood. Naked carbs are the devil.
Yeah, as I share in the article, my Thanksgiving meal had the same (or not more) carbohydrates than the oatmeal and my glycemic response was significantly lower, likely because it was a mixed meal with fat/protein.
Very interesting! If you had to hypothesize, do you think that women who consume higher amounts of naked carbs during pregnancy might predispose their offspring to diabetes epigenetically?
No need to hypothesize. There’s research showing precisely this. See Chapter 2 of Real Food for Pregnancy in the section on carbohydrates.
Thanks Lily, brilliant article and insight to your experience.
Personally before I was pregnant I followed a very strict Paleo Ketogenic protocol, 15 weeks into my pregnancy and replaxing my carb intake ive gained 12 lbs, I’m very interested in running this experiment on myself to see what I can and can’t tolerate well. Do you think being pregnant would throw off the readings, or wouldn’t make a difference?
I would be more than happy to share my results with you.
Thanks again Estelle (estelleskitchen)
Pregnancy tends to result in slightly lower blood sugar readings (see this article) and the “goal” blood sugar readings are lower than non-pregnant.
If you have my book, Real Food for Pregnancy, see Ch 9 for an in-depth discussion of blood sugar goals and different screening options for gestational diabetes.
Thank you Lily for insight and experience taking this test.
Personally I have been considering doing this myself, as for many years I followed a strict Paleo Ketogenic protocol and use to measure my BG and Ketones regularly.
Fastforward and I’m 15 weeks pregnant, relaxed my carb intake and concerned about the rapid weight gain of 12lbs. Thinking allowed this might provide me the tool to see what carbs and I can can’t tolerate well (very much inline with Robb Wolf’s carb test). Do you think being pregnant would throw off the results somewhat? I would be happy to share my results with you for your readers.
Thanks so much Estelle
Thanks so much for this great article! I have a history of GDM and will still occasionally check my sugars, but have struggled trying to figure out what is a truly normal response to a higher carb load so your research is very interesting!
I’ve read a little about “physiologic insulin resistance” in those who eat a lower carb diet (especially Keto) and I wonder how that plays into your spike from eating oatmeal? If you started eating more carbs daily would you spike less? For those that overall are eating a lower carb diet that is having good metabolic results what is the result/outcome of then the occasional higher carb meal (and potentially higher blood sugar spike)? Do we need to occasionally make our pancreas do a little work so it doesn’t forget how to do it’s job??
Yes, that’s the argument for carb loading prior to a glucose tolerance test, which essentially “primes” the pancreas to produce larger boluses of insulin. Technically, I had several instances of moderate carb loading — Thanksgiving, the Thai food with white rice — with significantly lower spikes than the oatmeal. Technically, my response to oatmeal would still not classify me as either prediabetic or diabetic because my blood sugar returned to “normal” within 2 hours.
It’s very difficult to find data on what’s “normal” or not in terms of blood sugar responses. Hopefully more CGM studies, similar to those I mention in the article, will be completed in the coming years. For now, what keeps me feeling well and keeps my blood sugar in check is moderately low carb, nutrient-dense, real food.
I guessed the spike! Although I did think I was pushing it. What’s my prize?
A bowl of oatmeal to celebrate? Haha.
A real eye opener, thankyou for the great article.
One of the doctors I work with asked my advice for a low carb/ real food dietitian for pregnancy and resources for a friend and I gave her your name. I hope she read your work!
Thanks for the referral!
Bello. I am new here but not new to Dr.’s work on the microbiome or diabetes 2 as unfortunately. we are genetically disposed.
My question is;!had you tried plain oatmeal(for sciences sake ) I would really be interested to see if the raspberries & honey were to blame or if it was truly the oatmeal. Also, what kind of oats; steal cut, quick oats, instant oats, rolled etc.
This is a great article. I’ve focused on eating protein, fat, fiber, and greens at meals (and keeping carbs moderate/non-starchy for the most part) for the last year… but now I’m traveling Europe and have noticed a STARK contrast when eating naked carbs (weight, acne, mood, energy). This article helped me to fully articulate the science of blood sugar balance to my husband. We’ll be using #nonakedcarbs as our reminder. 🙂 Thank you for your work!
It really struck me how much mindful eating observations were spot on to my blood sugar readings. Glad to hear #nonakedcarbs is working for you.
Hi Lily
Thanks for this fascinating insight, I was really interested to read it. I’m following your real food principles to gain control of my own gestational diabetes at the moment (doc’s say they think it’s actually type 2 but can’t be sure) and like many women was struggling with my first thing in the morning fasting readings. For a couple days now they’ve been normal. They were never in the normal range before, even with the slow acting insulin I was taking before bed, and even when this was dialled up slightly. I noticed it go down when I did two things: a) have a snack before bedtime and b) not take my prenatal multivitamin right before bed as usual. It’s probably the insulin kicking in and the snack, but do you know of any correlation between taking a prenatal multivitamin at a certain time and a spike in blood sugar?
super interesting Lily! As a fellow RD and CDE, I think you’ve inspired me to do this experiment as well. I specialize in PCOS and am very interested in a low glycemic diet. A few questions/thoughts: what type of oatmeal? I wonder what would have happened it you had used steel cut oats, added chia or flaxseeds and an ounce of nuts? (and not added the honey or fruit) Second, what do you think about Day Two? I realize it’s not even close to the CGM, but have you had any experience with it?
It was rolled oats. Years ago, I ate steel cut oatmeal for breakfast every day (as I explain in this post from my archives). I added all the “good” things to up the fat/protein/fiber. I didn’t get hungry quite as quickly, but felt just as awful. My expectations is my blood sugar probably wouldn’t spike as high, but it would still spike from a high carbohydrate load. If I ever get another CGM sensor, I may experiment with this more.
I loved reading this about this whole experiment! I would also be interested if you ever did more experimenting with oatmeal. I’ve been a type 1 diabetic for over 30 years, and before I ate a low carb diet, I would occasionally have oatmeal. For some reason, oatmeal (rolled oats, soaked overnight) would really spike my blood sugar more than any carb (rice, white potatoes, etc.), even when I added coconut oil to it. I have another type 1 friend who says the same about oatmeal. I’d be interested to see a non-diabetic compare white rice, white potatoes, and oatmeal (with and without fat/protein), or something along those lines. I’ve often wondered if there’s something about oats…
Thank you so much for doing this experiment and writing it up so thoroughly!
I have reactive hypoglycemia (non-diabetic) and oatmeal makes me feel terrible.
Great article Lily and hope one day soon the Libre will be available wo a prescription like glucometers are now. The glucometer and CGM are such powerful behavior modification tools as you demonstrate. I need to get one and wear it for the 10-14 days to learn more.
I hope so too, Mark!
Excellent article! Thank you for doing this experiment, all the added research and sharing it with us! I’m curious why the two week cut off? Perhaps that was all your doctor would write the script for? Anyway I was just wondering why you didn’t wear the CGM longer to be able to test more foods and food combinations.
You’ve inspired me for sure! Keep up the great work; I love it!
The Libre sensors automatically stop working after the 10-14 day cycle. I can get another sensor to continue measuring glucose should I run another CGM experiment.
Great article Lily, thank you! I was wondering if there was a specific brand of glucometer you use? I had a relion prime and it never seemed to read reliably. Thank!
I have the most reliable results with the Freestyle line of meters (no financial affiliation).
I would be curious to see how a dessert (higher sugar than oatmeal) would affect your BG. Do you ever eat dessert/treats?
Also curious about your usual physical activity level. It would be interesting to see how a day of just moving more (life chores/tasks) affects BG versus an hour of high intensity exercise followed by a full day of desk work (my typical day).
Thanks for your great n=1 research work Lily. Very useful for me because I am about to embark on a similar test with a Freestyle Libre (I’m also non-diabetic) so you provide really helpful benchmarks. I particularly like your sub-head – ‘Wearing a CGM cuts through all the noise’ – that’s the biggest plus, for us all.
My only concern is that this type of ‘individual’ approach gets used to reduce dietary generalizations and provide arguments for how different we all are, and how we need more ‘made-to-measure’ approaches to nutrition. While some individuals will be less responsive to oats or rice or potatoes, the essential principles advocating a lower-carb, real food diet seem to be the best basis from which to start.
Many, many thanks for your patient and complete approach… Sammy
Thank you for the excellent article. Very well written and SO interesting. Makes me want to do an CGM experiment on myself. Now if only my doctor would prescribe me one. 🙂
Hi Lily,
Thanks for the great insights.
As you are already low carb, it is not surprising to see spikes in blood sugar with even little bit of carbs (rice and oats) in your experiment. During low carb eating, glucokinase is down regulated and physiologic insulin resistance kicks in which means glucose takes lot longer to get disposed of as compared to someone healthy who is not on low carb. This is at least true for most low carbers including myself. An OGTT taken in this state looks scary. It would be very interesting if you ate 150g carbs per day for 3 days at least and then monitored your blood sugar response to carbs.
Just excellent! Thank you for taking the time to do this experiment and write up afterwords!
Hi Lily, I really enjoyed your post! Thank you! After reading it, it made me think of one of Chris Kresser’s post regarding low carb and pregnancy. I wondered if you had thoughts on the points he raised at the end when discussing the “Fetal Origins of Disease Hypothesis”.
For example, he writes…
The fetal origins of disease
“Nutrition during pregnancy is important for more than just supporting the growth and development of the fetus, though. It also programs the metabolism of our children for the world they will be born into.
This is more formally called the “fetal origins of disease” hypothesis, which has been well supported by the scientific literature. The most oft-cited example is children who were born to women during the Dutch Hunger Winter during World War II. Severe undernutrition of these women early in pregnancy produced children who were metabolically programmed for a world where food was scarce and where conserving energy and storing fat was advantageous. When the famine ended and food became plentiful again, these children were predisposed to obesity and metabolic syndrome in adulthood (4).
There are only a few studies that have looked at carbohydrate restriction and how this affects the health of offspring later in life, but in general, they don’t favor low-carb diets.
One study in humans found that the offspring of mothers who had consumed higher levels of protein and fat (likely resulting in lower carbohydrate intake) had significantly reduced insulin production in response to a glucose challenge 40 years later. For mothers consuming adequate protein, there was also an average 9.3 mm Hg increase in adult blood pressure for each 100 gram decrease in maternal carbohydrate intake (5).
A similar study found that a high-protein, low-carbohydrate diet during pregnancy was associated with increased cortisol levels in the offspring 30 years later (6). (Unfortunately, these studies did not report micronutrient intakes.)
Animal studies have suggested that a ketogenic diet during pregnancy may reduce the size of brain regions like the hippocampus in offspring, while increasing the size of others, such as the hypothalamus (7).”
Here is the link to his article from March 27, 2018:
https://chriskresser.com/do-low-carb-diets-during-pregnancy-increase-the-risk-of-birth-defects/
No worries if you don’t have time for this! 🙂
I encourage you to read chapters 2, 7 and 9 of Real Food for Pregnancy for an in-depth look at macronutrient “requirements” in pregnancy and the implications of elevated blood sugar as well as chapter 11 of Real Food for Gestational Diabetes if you’d like to understand the science for/against low carb in pregnancy.
Thanks Lily! I have your Real Food for Pregnancy book and have read portions, but will check out those chapters. Epigenetics is such an interesting field in relation to pregnancy. Thank you!
Super article. It reinforces my own experience as a T2 using a CGM. I am a 67 year old British man so not your usual reader.
However, I will also add that for me, wheat and oats are a one way switch. My BG rises to as much as 20 (360 American) and does not go down until the grain is expelled. Rice spikes a bit less and goes down. Even straight sugar is not so potent, although I have never attempted to eat a similar amount. Using cream as a source of fat moderates the spikes and eliminates hunger on the downslope.
Cereals have an enormous role in diabesity that is hardly discussed.
My usual breakfast since my CGM opened my eyes is grapefruit or microwaved blueberries with cream followed by two strips of bacon with tomatoes/avacado/mushrooms/baked beans.
Fasting until about 13:00 also avoids huge spikes as insulin resistance seems to decline over the day. A large roast dinner has almost no impact on my blood sugar.
Great writing too.
Thanks for sharing your experience, Phillip.
Interesting results. Could you elaborate on the type of oats you used? Was is the plain, large chunks (which you have to boil for ca. 15min) or the express type (done in 2-5min)?
Old fashioned rolled oats (not quick cooking; the kind that take 20+ minutes to cook), organic, certified gluten-free.
Perhaps if one adds some whey protein and some fat ( eg nut butter) to the oatmeal the response might be better?
Likely yes, hence the #nonakedcarbs discussion. The meal I tested was specifically to mimic the sample meal plan from the Academy of Nutrition and Dietetics (oatmeal, milk, berries) and see what happened with a meal of primarily carbohydrate.
Interesting!
1. For the geeks in your audience, it would be nice if the references were more specific so people could read them. “JAMA, 2016” isn’t too useful.
2. JS Christiansen of EASD in 2006 used CGMs on allegedly nondiabetic people after a high-carb breakfast and reported the results at a conference that used to be online at diabetes-symposium.org, but I think no longer is. They showed individual results, and some people didn’t go over 100 at the peak whereas others went quite high.
3. I also get ravenously hungry when a peak starts coming down. The analogy I use is, if you had a toddler coming down a slide and there was broken glass at the bottom, you’d run to stop the kid *before* she reached the bottom rather than waiting until she was in the glass. That’s what the body is doing.
Sciarc.
See the references tab at the bottom of the post for full citations.
Lily,
Very informative post thank you.
I’d like to suggest 4 extra CGM tests:
1. Measure the effect of 2 measured glasses of dry white wine, one before a meal and one with a meal. (100 ml is a standard serve, but 150 ml is a restaurant serve)
2. Measure the effect of a sugared soda with a meal.
3. Measure the effect of a diet soda consumed alone when fasted. (I expect there will be no blood sugar response, but may be wrong. But I would expect that there would be an insulin response, which a CGM would not pick up – I think.)
4. Whether reduced sleep (say 6 hours versus 8) affected the response to a breakfast.
Best wishes from Brisbane Australia
Jamie, you mention doing a test with white wine. In my limited experience, I find any sort of alcohol before food and there is a very good chance I’m going to have a hypoglycemic episode, most times even after I have consumed a well balanced whole food meal. Other than skipping the alcohol, any way to maybe stop this from happening? Many thanks!
I don’t think you’re gonna get this chick to drink a ‘sugared soda’
Quote ‘This data highlights the role that excess body weight plays in blood sugar regulation, even if you “pass” diabetic/prediabetic screenings by conventional standards.’
Or could this observation show raised blood glucose; spiked insulin leading to fat storage & weight gain ie being over weight was the symptom not the cause!
Great article and experiment!
I was wondering what it was like to remove the sensor after your experiment was done?
Also, the earlier comment where the gentleman recommended having you drink diet soda would be really interesting! You might be able to experiment with other no calorie sweeteners like Splenda, Truvia, or monk fruit and see your body’s response to them.
Removing the sensor is like taking of a band-aid. You do not feel the little, thin plastic thing that goes under your skin. Not when you stick it on and not when you take it off.
This is fantastic Lily! Well done! Great to see this in action!
Happy new year and keep up the fab posts! 😉
Andrew
You go girl! THanks for putting yourself out there. I literally read this while my in laws were in town. I had just told my MIL that her blood sugars were in prediabetic range and we may want to rethink how she starts her day (homemade muffins and oatmeal). I was glad to share with her your results. It’s amazing how people don’t think they can get fiber or have normal bowel movements without oatmeal!
This article is super validating. I can’t eat something like oatmeal or toast for breakfast because I’ll feel ravenous within a half hour. However, I can eat an egg with spinach or a piece of buckwheat toast with butter and be satiated for hours. Thanks for your experiment!
I am wondering about alcohol consumption, which includes wine with dinner, or a glass of wine with hubby in the evening after a tough day, etc. Some think this is beneficial to health. Is it the combination with good food that makes the difference? What do you think?
Yes! I Thank you for this post. have been wondering about all these things. Thank you for doing this experiment—we definitely need more data on glycemic response in healthy people. I am also very interested in how the fourth trimester affects glucose response in healthy individuals vs those with GD, particularly in the initial phases of lactogenesis. I have yet to find much data on that either, but I suspect that the 24-hour feeding cycle (ie interrupted sleep) plus all the hormonal changes happening during that time must impact insulin sensitivity. However, the typical medical advice seems to go only so far as to say that “usually glucose levels go back to normal after birth”. I would love to see more data on what is actually going on in the post-partum breastfeeding momma. Thanks again!
Randy Woodward So, there is a VERY important lesson to be learned here, which I have confirmed myself. On the oatmeal experiment, recognizing how much her blood sugar level spiked is one thing, but the more important thing was what followed later…”What was interesting, though, is that I got ravenously hungry when my blood sugar started plummeting.” What this means, and I’ve confirmed in my own experiences, is that even if you are a full-time low-carb eater, which allows you to access your stored fat for energy, which is ultimately the goal here, THIS ABILITY WILL IMMEDIATELY CEASE WITH ONE SERVING OF “NAKED CARBS”, as she puts it. With a good low carb diet, I can ride my bike for hours on end. As a matter of fact, I did the Leadville Race Series MTB 100, that stands mountain biking 100 miles at a 9,300 to 12,500 altitude, in 11.25 hours. I woke that morning and had eggs, sausage, and bacon. During the race, I had ONE protein bar, more so out of boredom, than need. And of course lots of water. Think about that! I had to burn 8 to 10k calories, and I had maybe 250 calories during the race. By the way, Gary Taubes taught me this could be done in “Why We Get Fat”. Anyway, so one day I decided to experiment on a 50 miles ride, that would last about 4 hours. I ate my no-carb breakfast as I always do, and set out on the ride. At the 1 hour mark, I stopped and had a donut and some orange juice, and set off again. Within an hour I completely BONKED! Which is exactly what Lily described…your block sugar level tanks, leaving you know carb energy, and your access to stored fast has been SHUT DOWN! It happens that quickly and that easily. Period! I’ve tested this theory several other times and in different environments, and it happens every single time.
It’s NOT what were “told” about the benefits of eating low-carb that make it real or truthful, it’s about what we actually do and feel to prove to ourselves that it’s real and truthful.
Interestingly, Atkins referred to this without really knowing why, at the time. He made a comment in his book that when you’re going zero-carb for a period of time, even a mint or a stick of gum can throw the whole thing off.
Thank you for doing this! I would be interested in reading more if you ever experiment more in this way. I’d also love to see some continuous monitoring before and during the 3 hour GD test. For me, my goal is to pass the test (which I have always done, but sometimes barely) and then self regulate with a gd diet without a diagnosis or finger pricking. ( I have used three different home monitors and tested them against my blood results. EVERY SINGLE TIME my home moniter reads my fasting and post meal readings 20-30 points too high compared with my blood that was sent off to the lab, taken at the same time. So if I get diagnosed with GD, my provider will look at my finger prick numbers and say that I can’t control with diet and need meds.) Anyway, my ‘hack’ for the last 2 pregnancies has been: low carb, then super high carb for 2 days, than a day of super low carb and a ton of exercise, take the glucose test, then go back to a low carb diet. I dont know if that’s all necessary, so I’d love to see it tested changing each of the variables leading up to the test! Even better, I’d love to see the test changed to a not naked carb intake!
Hi Lily,
Thank you for taking the time to write up this piece and make it available for us. I am a Type 1 of 16 years, and I have struggled to find much useful cgm info from non diabetics (specifically individual day charts). Your oatmeal experience, especially the notes about being very hungry during the falling glucose, regardless of the actual level is a terrible phenomenon that we Type 1s deal with often. Unfortunately we are taught that we can eat whatever we would like as long as we cover it with industrial doses of our fast acting (actually very slow) meal time insulin. This behavior creates a roller coaster effect of levels that induces a lot of binge eating because of that feeling you experienced. I have just recently begun to be a low carb eater and get my cgm data to look a lot more like your normal data. I have always wondered what that maximum spike a non diabetic might experience when eating heavy carb foods, and the oatmeal chart was exactly what I have been looking for.
Thank you again,
Cheers!
Fascinating, enjoyed the breakdown of your experience. In regards to your spike in blood glucose after the standard american breakfast, I’d imagine that’s to do with your low carb adaptation. If you’ve eaten mostly low carb for the trailing ~14 or so days (or even the past 24-48 hours before the meal really), you wake up fasted, and as a low carber have physiological insulin resistance, and slightly higher circulating ketones. On eating oatmeal this would have driven your blood sugar to multiples of that of a normal person who wasn’t on a low carb diet. If you at moderate amounts of carbs for three days in a row, and repeated the experiment, I’d guess the spike would drop down significantly. (same reason why you have to eat a significant quota of carbohydrates in the days prior to the OGTT for it to be statistically valid.)
That’s an interesting theory. I have been doing CGM as a non diabetic and was trying to benchmark my results against others, which is how I ended up here.
I eat 40g of porridge oats every morning. I eat them with a heaping tablespoon of peanut butter (actually closer to 2 Tbsp) and a sprinkling of seed and dried fruit mix. I pour boiling water over the oats and stir all of that together but don’t otherwise cook them. For my part, I never put sugar or honey on oats and indeed living in Scotland, where people are more likely to put butter and salt on their porridge, I’d be laughed out the room if I said I had, LOL!
Looking at my week, with the above oat / PB / seeds combo, I have never had a spike over 100 mg/dl and on days when I’ve not had a (oat)milky coffee alongside it, my glucose level barely budged. For example on Sunday it looks like it was only up to between 70 and 80 just after and for the next 2 hours on the oat breakfast described above and no coffee with oat milk. I eat no added sugar and no naked carbs, otherwise a plate that’s half veg and the other half split between protein and complex carbs. So perhaps my body has adapted to that more moderate intake and thus isn’t overly fussed by my morning porridge?
Thank you Lily! As a fellow RD who also has a low carb philosophy, it is great to read blogs like yours that are helping educate folks and hopefully get them on a healthier path! Keep it up!
Glad you enjoyed reading about my continuous glucose monitor experiment, Sandra! Cheers to more real food and low carb friendly dietitians. 😉
Great work Lily. Thank you for challenging the status quo and for your investigatory mind. YOU ARE AWESOME and changing dietetics. Hugs 🙂
Thanks Tara. You’re right there with me helping to move dietetics forward. 😉
Excellent self research! I have had a similar experience with testing my own glucose levels with a glucometer, especially with rice. I have 2 deceased parents with type 2 diabetes and am prediabetic myself. I am an almost 70 senior female and would love to try CGM myself. My daughter in law is type 1 and wears one and a pump and it has made a huge difference in her monitoring. Thank you for this self reporting, we need to see more of this!
I am non-diabetic who tests my post prandiel blood sugar after meals for fun, well initially in an effort to lose weight and because I am seriously obsessed in my own data. I ate low fat, high carb for years because my father died from a heart attack and that was the recommendation. I have the same experience with carbs meals. My worst meal ever is a bagel, and it doesn’t matter if I slather it with cream cheese or put egg and sausage on it. I was even able to eat a donut after having a bowl of cottage cheese with less of a spike than the bagel. BTW I lost 13 pounds of post baby weight that I couldn’t lose for 15 years by ditching added sugar foods and reducing grains to a small portion a few times a week. I think your advice is spot on.
Hello Lily,
Good article. I am a Health Architect from the Netherlands and have been testing the device five times. Next to see what food is doing to your blood glucose levels, you can als see the effects of physical actions such as workouts, running, etc.
What also is surprising is what taking a sauna is doing for you blood sugar. Actually it shoots your BS up to 200+!! It also falls back to normal levels pretty quick.
With regard to your Thai food experiment I have my own observations. If I order (Chinese, Thai types) food from restaurants I will see a huge rise in BS even if I don’t order rice or noodles. I first thought it was due to a lot of sugar in the sauce. However, I after investigating it was not the sugar but more the MSG (monosodium glutamate) used to flavour the foods. Just imagine what happens if you are diabetic and go out to eat this.
What is your experience with this issue?
Interesting, thanks for sharing your thoughts. I know a lot of Chinese food uses corn starch as a thickener in the sauces, so even if it’s not sweet, it has hidden carbs. I’d be curious to see how pure MSG would affect blood sugar levels, but I’m not about to sign up to test that out!
Whoa! Lily. What a shock on your results over the oats! I have been making oat pan bread in the morning with only turmeric and water added, for my dad. (Plus giving him a drizzle of coconut oil over top as well.) He just moved in with us and was previously constipated all the time until he began eating this. He’s clockwork regular ever since. I eat it now too because it’s so good. But NOW I am going to add egg to the mix. Why? I was really wondering why I was feeling weak and hungry all morning after that oat bread. RAVENOUS by lunchtime. Of course! Carbs, regardless that they are laminated (rolled) oats. I figured: Hey. It has fibre and will slow down the digestion. Thanks for this eye-opening post. Glo
So informative. Totally want to get my hands on a CGM. I did Robb Wolf’s carb test on bread, potato, white rice, oatmeal, and banana. The only one that didn’t spike my blood sugar over 130 after an hour was the bananas. All the others pushed me to 180+ and just like you, starving! Explains so much of my childhood to college years and feeling so hungry all day.
I loved this article ! So much information! I wanted to ask you, for a pregnant woman struggling with very severe constipation and wanting to increase fiber content, without resorting to oatmeal and grains and high carbohydrate fruit (too much of it), what are good options? Thank you in advance
Thank you for the writeup. I’m not sure I would have had the nerve to try a CGM experiment without having read this post.
My father has longstanding (20+ years) T2D, gone insulin-dependent after a couple of years, and T2D runs in his family. I’ve been low-carbing since the South Beach Diet became popular, and exercising regularly since my early 20s; having seen what Dad deals with, I don’t want to end up T2D at 60 years old like he did.
Dad got a FreeStyle Libre 10-day last spring, and has loved it. It suits him a lot better than a smartphone-type CGM would–he doesn’t do computers, even little ones. He upgraded to the 14-day Libre last week, and still had one 10-day sensor left, so he gave me the 10-day reader and sensor.
Three days into the sensor, I have a good idea of what my “normal” is, and I’m about to start experimenting more extensively.
My husband is also T2D, on meds only, moderately low-carb, doesn’t exercise, and rarely checks his BG. He’s open to the idea of getting a sensor prescription from his doctor and giving it a try. I believe it would be an eye-opener for him. It already has been for me.
Glad to hear you got your hands on a Libre for your own CGM experiment! It really is an eye opening experience.
Excellent write-up! I wish these things had been around fourteen years ago, I had to work out what was happening longhand with a glucometer.
I discovered that my glucose would spike around 1 hour postprandial, be almost back to normal at two hours and then at three-four hours it would drop low pretty much as a reciprocal of the spike. Not just the low but the speed of the drop was what brought on my (reactive) hypo symptoms due to the release of counterregulatory hormones, not just glucagon but cortisol, epinephrine, norepinephrine etc. Like “hangry” on steroids.
I found the same as you with those “healthy whole grains” – wheat was my kryptonite, the only thing that spiked my glucose worse than wheat was wheat mixed with other carbs – I suspect wheat germ agglutinin – and curiously when I gave up wheat I mostly stopped farting and my GERD resolved. Who knew? Well lots of people actually, just most of them aren’t doctors.
The other important thing I discovered was the pattern of insulin resistance being highest in the morning. When I started I was limited to about 15g carbs at breakfast and 30g by evening. Once I got my insulin resistance under control my breakfast dropped to about 10g but by evening I could eat 50 – 80g and sometimes 100g without spiking. Not that I do but I CAN – mostly I have stuck to around 50g carbs mainly in the evening. This pattern is common in Type 2s but many Type 1s have the opposite pattern. Non-diabetics? Who knows, are there many left?
Seriously though I think the CGM is a game changer, there is a strong financial case for giving every diabetic and prediabetic one on loan for a few weeks now doctors are finally permitted to believe diabetes is “reversible”.
Already though I have seen clueless dieticians (other types of dietician exist, obviously) claiming that it is dangerous to use a CGM because you may give up “healthy foods” like cornflakes and fruit juice just because they spike your glucose. One such claimed that since he found glucose spikes after breakfast, that just meant that “nondiabetics like him” also had glucose spikes. Ermmm, you may not be diabetic yet but you are already fat, just maybe the CGM is telling you something . . .
A recent JAMA paper
Assessment of a Personalized Approach to Predicting Postprandial Glycemic Responses to Food Among Individuals Without Diabetes
hopefully more such research will be done to back up all the “just anecdotes”
What concerns me is that all this religious monitoring of glucose levels seems to begin with the assumptions that:
– all glucose peaks are bad
– all large glucose peaks are the fault of the carbs
Regarding the first assumption, what is the evidence that momentary glucose peaks are bad for health? And to be clear, I am referring to people who are not overweight and already in a disease state.
Regarding the second assumption, I see no where in your write-up where you are considering non-immediate effects of your diet on your instantaneous glucose readings. There have been studies going all the way back to the 20s that show pretty clearly that fat consumption can have an impact on glucose response and insulin resistance and yet you seem to be completely ignoring that. It seems to me that if we ignore the non-immediate impacts of diet we are in danger of doing the equivalent of determining that the pain in our leg is caused by running or walking instead of the sprain we suffered days previous.
As a side-note, I don’t find your comments on your “feelings” after a certain type of meal very compelling. Those “observations”, particularly on N=1 studies like this, are ripe for confirmation bias. I think the controversy over the sugar/hyperactivity relationship in children and the fact that many people on a whole-food, plant-based diets DO NOT experience shakiness or roller-coaster energy levels is enough to at least question whether your “feelings” are legitimate for everyone or even a significant portion of the population. There is also again the possibility that your “feelings”, if relevant and un-biased, are reflective of your diet as a whole and not just what you ate at your last meal.
Lily,
Great article, second time to read it. I saw you presentation at Low Carb Denver and now ‘experimenting’ with my CGM (I am non prediabetes) to see how accurate and correlate with my LCHF diet.
My CGM (FreeStyle Libre) was showing 25 point higher than my KetoMojo blood control, not sure if is a sensor issue (poor answers from Customer service).
I am a Surgical Oncologist, super interested in Keto and Cancer. But we need the basics of DM control first, horrified with my patients poor control and lack of diet knowledge.
Reach out to Abbott. That sounds like a faulty sensor.
Perhaps your oatmeal experiment had everything to do with the added HONEY. Rolled oats leave much of the germ behind, leaving mostly endosperm vs. steel cut oats. Rolled oats also have a higher glycemic load and less fiber than steel cut. Perhaps steel cut oats with only berries would not have had the same effect.
To blame a 100-point spike solely on only the 5 grams of carbohydrates coming from honey (while ignoring the 40 grams coming from oatmeal + milk) is a far stretch in my opinion. I agree that it *contributed* to the spike.
I really like reading a post that will make people think.
Also, thanks for allowing for me to comment!
Just an FYI, the Libre tends to read low, especially at night. They actually have a disclaimer about this. Also, not sure how low-carb you typically eat, but if you primarily eat low-carb, a sudden influx of carbs will cause a bg spike as the enzymes needed to handle a carb load are made on an as needed basis, but there is a delay. It takes about 72 hours to start making them again, which is why it is recommended to eat at least 150g for 3 days prior to an OGTT. A low-carber will fail an OGTT even if they are not diabetic.
My CGM was within 5-10 points of my fingerpricks, though I have read in the literature and seen clinically that the Libre can read on the low side.
I eat moderately low carb, not keto. The oatmeal experiment was a few days after Thanksgiving (a day which invariably had a LOT of carbohydrates). Even Thanksgiving dinner didn’t spike me as high as the oatmeal, which is likely because it was a mixed meal with plenty of fat and protein. It’s possible oatmeal is an outlier for me compared to other sources of carbohydrates, just like the CGM experiments suggest.
Thanks for sharing this. I’m a 42 years old man and was diagnosed with T2D with an A1c of 12.1% this xmas. I have been wearing a Libre CGM on and off and made significant changes to my diet (I still eat carbs but more moderately usually around 30-45% of total calories–and trying to focus on whole grains and vegetables) and added some exercise. I’m also a vegetarian. It’s super helpful to me to have data from non-diabetics to compare to as I’m trying to get my own diabetes into remission (my last A1c was 5.2% so I’m making good headway :-). So thank you so much for sharing your experiences and a little bit of non-diabetic raw data. A few comments on my experiences.
1) Oatmeal causes a big spike for me as well, it’s almost exactly the same shape/profile although actually not usually as high. This was a big surprise to me as it was my breakfast of choice before wearing the CGM. I’ve switched mostly to Chia seed pudding now.
2) I’ve only worn a few sensors so far, but have read obsessively in diabetes forums on other’s sensor results and so far my experiences have been similar.
a) The Libre sensors can vary quite a bit.
b) Most sensors tend to error low most of the time (compared to pin pricks), but some “hot” ones error high in the mid range (my current sensor often puts me 15-20 higher than pin pricks on average–my last one put me 10-15 lower on average). (I have 4 meters and they vary some amongst each other too–but my AccuCheck Guide and an ContourNext One are usually pretty close to one another and pretty consistent with multiple tests in the same time period)
c) It seems many report it’s fairly common for the Libre sensors to report errors at the extremes in both directions (highs too high, lows too low). This is also consistent with my experience. That might be a deliberate calibration for safety?
3) It’s getting harder as the 14-day sensors now encrypt the data and don’t work–but there’s fascinating 3rd party products (search BluCon or MiaoMiao) that continually scan and relay the data to phone from the older devices, and 3rd party apps like xDrip or Spike that can perform additional calibration with pin pricks to improve Libre accuracy. Fascinating how active the diabetes DIY community is. I haven’t been able to try these as I have a 14-day sensor/reader.
I also appreciate Daniel’s comments above. I find it’s easy for me to start villanizing all glucose spikes (and that conclusion doesn’t seem entirely unreasonable), but I’m not sure there’s sufficient data to really support that. I do find that most large peaks for me do seem to center around carb consumption though (although other factors do also affect me, particularly sleep).
Today I ate one of the largest carb meals I’ve eaten in a long time–a potato breakfast hash that I estimated at 121g of carb. Surprisingly rather than causing the massive spike, I’d anticipated/feared–I ended up with a prolonged high (120-135) for around 3 hours. This may support your idea of not eating “naked” carbs–as my hash included eggs, cheese, and oil as well.
Cheers!
Yep, I have read some small case reports in the literature of the Libre showing extremes of highs or lows (higher highs on a glucose tolerance test, for example). Mine seemed to match up pretty close with the finger pricks though.
Glad to have found your blog. My response to keto is similar with a CGM. A stable line with little variation around 65-70 in winter and 75mg/dl in sommer. It took me about 2 years to get there. Now I can tolerate some fruits without spikes in BG. Spikes up to 90mg/dl happen only with more exercise. Thankx for sharing!
I have just been wearing a cgm for fun and was horrified to see my glucose go to 11.2 mmol after some oats ! I am on a low carb diet usually and worried this has ruined my carb tolerance but reading your blog has reassured me it’s the oats! That’s how toxic they are!!
A really well thought out article that I thoroughly enjoyed. Good information combined with anecdotal/real life experience, relevant citations and studies… the quality of which you don’t see often. Thank you.
What a great article! I plan to share this with my midwives in hopes that CGM becomes an option in place of the glucola test. I wondered if there’s any research or discussion on how breastfeeding effects blood sugar regulation?
I am pregnant with our 3rd and currently breastfeeding and also breastfedthrough the 7th month of my second pregnancy. My starting weight was higher with the second and I did pay more attention to carb intake however, my weight gain was about 30lbs vs about 50 with the first. I would try a CGM myself, out of curiosity and a desire to prevent disease!
I need to get your book and read it! Thanks Lily for all the work you do!
Check out Dr. Ken Berry’s wife Neisha. She has a YouTube channel where she talks about carnivore diet, pregnancy, “gestational diabetes”, and breastfeeding.
Hi Lily, Feeling confident over FSL after reading your article. It was such great help. I will also buy it for my diabetic father-in-law but I have couple of questions before buying Libre. Would appreciate if you could help me answering them so that it would easier for me to decision for his health?
Q 1) Does FreeStyle Libre a waterproof sensor?
Q2) I have read in many blogs about the Ambrosia’s nightrider blucon. I don’t know much about it but bloggers says it’s a third party device which sits on the FSL and gives reading to mobile every 5 minutes. It’s strange that people not only in US are using it but it’s being used almost in every part of world. Moreover I have read it gives high and low alerts, caregivers can follow readings too. Though It’s good to hear that we’ll be notified about highs and lows but is it actually true? Have you ever heard about it?
FreeStyle Libre is waterproof enough to take a shower. They advise you not to stay in water longer than 30 min though.
Who paid for the testing and time to do it?
I explicitly explain in the article that I paid out of pocket for my CGM experiment with the full costs listed.
I find that at the lower end, the Libre reads about 1mmol/l (18mg/dl) lower than my Suresense blood glucose tester. It would be interesting if you took the Libre Hba1c estimate and compared it to an actual lab test result.
Hi Lily
Enjoyed your article – thanks. Found after trying to find something to compare my n=1 results.
I’m a 52 yr male, 1.84m, 76kg, mostly carnivore when by myself, but add other foods when in company; in and out of light ketosis easily both ways.
Have been wearing a CGM (Dexcom G6) for the last month. Very similar conclusions. Highest reading was 7.5 mmol/L after an experiment with some chocolate cake and ice cream, and lowest 2.4 on the rebound. Usually steady between 3.9 and 5.6. (average 4.4), with other the higher numbers due to exercise, relatively poor sleep, and dealing with characters at work.
The G6 was accurate for 2/3 sensors (within 5% of an averaged two reading finger-prick), but one sensor needed calibrating twice daily. Good app and associated summary / analysis. Very easy to insert and remove.
The ‘so what’ to all this must be inclusion of CGM as a key component of diabetes (etc) management. For me, it was interesting, for others it could help change everything.
Thanks again. Nick
In my experience, the Libre reads falsely low, especially at night. I’m curious what meters you tested against as the Freestyle meters also tend to read low.
As far as the oatmeal response goes, when we eat lower carbs, our bodies stop making the enzymes needed to handle the carb load. They are made on an as-needed-basis, but there is a 3 day lag, which is why they tell pregnant women who are going to take an OGTT to eat at least 150g for three days prior to the test. BG’s will spike into diabetic numbers if high carb foods are eaten after being low-carb for a while.
Hi Lily,
I’m so happy I ran across your article. I’m currently in day 3 of my CGM (freestlye libre). I’m a certified nurse-midwife, 14 months postpartum from a GDM pregnancy too. I used a CGM in my pregnancy because 1. I thought it was superior to finger sticks. And 2. I was motivated to not go on medications.
So anyways I popped a CGM in this week to see if I could handle a vegan diet. The answer to that I believe is no, I can’t. Like you describe it, I had a dinner full of naked carbs. I’m at 2 hrs post prandial with a level still hanging out at 150.
Thank you for your post I needed to read that there is a lot of nutritional jibber jabber out there that was making me think I needed a more plant based diet but the moral of the story is my body can’t handle it. I believe I eat very healthy with lean meats, fish, vegetables, and whole grains.
Back to that diet! Thank you.
I like all readers comments enjoyed this experiment and reading about your results. Why hasn’t any manufactures developed a way to track insulin levels if it is linked to storaging fat cells? Can you explain the HOMA-IR test?
I have started wearing a FreeStyle monitor, too. By the way, you only need to buy the gluscose monitor to stick on the skin. You do not need to buy the device to read the data, because with the FreeStyle App your mobile phone will be able to read the sensor directly. And at least in Germany you don’t need a presciption from your doctor, you can just order it online directly from the manufacturer (I bet they also ship overseas). I had some pleasant and unpleasant surprises: Dark Chocolate (95%) has no effect on my blood sugar (hurray!), sweet potatoes go through the roof (over 200). I am now experimenting combining sweet potatoes with butter and see what happens. I would love to get more data on what is “normal” or “healthy” in non-diabetics.
I’m doing the same – its fascinating
I just tested it: When I have a large serving of sweet potatoe (mashed) without anything else, blood sugar shoots to over 200. When I mix 80 gramms of butter into the mashed sweet potatoes (about half a cup, a lot, I know) then my blood sugar only went up to 110. I also checked if Abbott will ship the FreeStyle from Germany to the US and unfortunately they don’t.
Wow, Interesting. I eat oatmeal for breakfast almost every weekday. On the weekends, I eat a similar breakfast to what you eat…eggs, vegetables, and some type of meat due to my wife wanting to eat that stuff.
I always feel terrible after eating the weekend breakfast but feel great after eating oatmeal. I exercise 5 to 6 days a week and I like to eat a bowl of oatmeal in the morning prior to exercise. I can run with no real drawbacks from the oatmeal. A common routine is Oatmeal, 20 minutes later I run outside for 60 minutes, do pushups/pullups during the run..come back – make a smoothie of berries, spinach, and some whey. I am usually good for eating until the early evening with that diet. I may eat something else towards the afternoon.
However, my point is that eating oatmeal often has really helped me control my weight and gives me the energy I need in the morning. I do use about a half TBSP of brown sugar(organic), 1 TBSP Flax seed, a handful of almonds and walnuts, cinnamon, and a dash of whole milk. I love it. I don’t know how I could go on without it in the morning! Now you have me questioning myself!
Loved your article. I am a relatively healthy 88 year old female with a HgA1c or 4.5, fasting in the 80, post prandial usually in he 110 (2 hours) if I eat carbs. I stick to very low carb, mostly veggies, small amount of berries, apple peels, kiwi. I do add 1/2 cup of beans sometimes. I am glutenfree, so that eliminates breads, cookies, rice…. I do check my blood sugar often, mostly 1-2 hours after meals and learned which kind of carbs to avoid. Thank you for reassuring me that I am doing the right thing.l
Hey thanks for that article. It was so informative. I’m a dietitian with a very similar diabetes history as you. I’m currently experimenting with a CGM.
Took a look at your facebook and found your focus on GDM, in that case, you need to be aware glucose data from GDM is quite lower than normal person during the pregnancy. Don’t use the same standard.
As a certified diabetes educator, I can assure you I’m well aware of this.
Great
Hi Lily, thanks a lot for that article! I’m dealing with my pre diabetes since 1y, wearing a CGM since 0.5y, and experinced only limited knowledge about this technology and it’s data amongst the doctors I can access (I’m a foreigner living in China, btw CGM is freely accessible here). Your article will help me a lot to steer my nutrition, behavior and medication in a reasonable way. I’ll try to follow your publications.
Hi lily,
I did my 1 hour glucose I was on keto for 1 month prior and failed my test at 151. I’ll go back for my 3 hour when my sinus infection clears up but my doctor has be convinced that I have GD because he said I should’ve passed the test if I was on keto and since I didn’t pass I most likely have GD. Of course I have myself believing I do now. I got a glucose monitor quit keto for now and my fasting level in the morning has been between 65 and 84 after 1 hour my levels are around 130 or less after 2 hours 125 or less. I would love your thoughts.
I should clarify 1 hour after breakfast, lunch and dinner and 2 hours after are those levels stated above.
I ordered a reader and sensor without a prescription. Just search online.
Meg, could you please give some links here to websites where you found the CGM for sale to a non-diabetic without a prescription, here in the US? I’d really appreciate it! Thank you.
Interesting article, thanks. Do you think it’s important to measure the ketone bodies in the blood? If so can this be done with the device you had with continuous monitoring? Or does one need to buy a separate device with strips?
In your article you said
“the hunger trigger (for me, anyways) is in response to the impending crash”… Why does that happen? I am thinking I’m reactive as well and as I make the peri-menopause to menopause transition, it seems like it is becoming a daily occurrence. I have been increasing my protein at breakfast because it was pretty nonexistent before, and including a protein snack 2-3 hours later but then an hour and a half later I’m starving. So confusing!
This is an amazing blog especially with details on blood levels of indigenous diets.
Excellent, informative. I’ve been eating low carb, whole food, and exercising for two years and would like to know in detail if this is working as I hope. Thanks again.
Hi Lily, great blog post! So into it. I do have a question though—what does it mean for fasting and postprandial insulin levels to be low/ideal, but blood glucose to be high?
Just as an example: If someone had a fasting insulin of ~4 and a 2-hour postprandial of ~30 after a glucose test (all good), but then had a FBG of 90-100 and several readings in the 150-180 range after eating minimal carbs. Wouldn’t insulin also need to be high to confirm insulin resistance, or is this also a marker of pre-diabetes?
Thank you! This is all so fascinating.
I really love your articles and your work. A question I have is, what do you make of a lower than “normal” insulin level?
Hello, and thanks for this.
Please bear with me as this is important for me and I need to understand the mechanisms.
The carb content of the rolled oats I buy is 57%. I use 90g (ie 51g carbs) in a 1:3 part recipe, iow 270g water + 90g oats = 360g total. The resulting mixture contains 14% carbs (51/360). For a portion size of 140g this results in a carb content of 20g. (I also let is stand overnight and only reheat in the morning for breakfast.) Nothing is added to this.
Apart from measuring it with the BGM, any opinion on the nett effect of this 20g of carbs? (I still think it is way better than (almost all) of the other highly refined cereals but I need to understand, as it will give me insight for changing dietary habits. Yes for sure bacon and egg trumps it all, but unfortunately not always possible / practical 🙂 )
Many thanks
Hi Lily,
Thank you for this article, I really enjoyed reading it. My son was diagnosed with Type 1 diabetes (23rd May 2020). I have slowly been transitioning to a low carb high fat eating lifestyle for my whole family since his diagnosis. I believe it can help stabilise his blood sugar and in the long run is a better lifestyle choice for my family. I purchased the Freestyle Libra to see how his food affected his sugar levels and also found oatmeal shot it up very quickly. I am getting a lot of noise from my sons doctor and his dietian in the sense that I need to give him more carbs as he is a young child (11 years old). Do you follow a low carb and eat well regime with your children? My priority is my sons well being and for him to continue to grow on his growth curb as he was prior to diagnosis. Do you have any advice for me, it has not been easy this past week and a half and I sometimes feel really overwhelmed.
Follow your gut! Our 9 year old son was also just diagnosed Type 1 and my husband has been type 1 since age 9 as well. I am trying to do the same thing, focus on low carbs with lots of healthy fat since carbs are what spikes blood sugar and fat helps to slow it. Anyone would benefit from only eating nutrient dense carbs when they do eat carbs. Most carbs are processed and full of unhealthy ingredients that certainly could not be argued to promote health and growth. There are tons of wonderful recipes if you search for anything keto.
So glad you did this! I just wore an IPro CGM (Australia) for a week. I had really good levels a part from 3 instances where my levels were
6mmol/l, 6.1mmol/l & the highest at 6.3mmol/l before bedtime, all those days we had a late dinner (so really 2hrs after i ate). They didnt let me get a good look at the report but i seen a “flag” of 3%(???) My fastings were all >4.8mmol/l (mostly around 4.2mmol/l), Ha1c 4.2, previous fasting blood tests >4.1 (at 6wks gestation). I do have a history of LGA (#1 8lbs 2, #2 9lbs 8 & #3 10lbs 10) no previous history of GDM. With this pregnancy i have Polyhydraminos & another LGA baby (estimating over 11lbs). I did opt for the CGM in case i did in fact have GDM. Since wearing the CGM i have been tagged as having GDM, they want me monitoring 4x day and go into to see them for Diabetes education. From everything i have seen my levels are good?!?!. I would love if you could point me in the direction of further research or credible sources i could look up, to try and educate myself. (I do have your book real food for pregnancy. The most important thing i think for management of true GDM is for better outcomes for mum & baby…i am failing to see how, with my levels, any management would result in better outcomes! All i can see is this will stress me out (3 other kiddies to look after too), pressure for induction early & heavy opposition for my planned home birth. I trust your guidance (& my lovely midwives) more!!
Brilliant. I also would really be interested in the levels in modern hunter – gatherers. I watched a movie game changers and also read a bunch of oppositional positions and it’s just really hard to know what’s true.
Interesting, being on a moderate carb, moderate protein diet did not do me any good. After eating more carbs now I sleep better. PMS is better. I sometimes eat oatbran porridge for breakfast but I put in nuts and collagen powder. I have no issues keeping full until 1 or 2 pm. I started a moderate, lower carb diet( eating between 80 and a 100grams a day) because I was diagnosed with insulin resistance. Skinny though, and normal visceral fat (I am weird like that) The insulin resistance did improve, I have to say. But my sleeping suffered, and I got severe pms. Now I am fine. I am on thyroid meds, Eltroxin, which is well controlled but never ever also had a problem with my weight. I have no thyroid. I wish I could just be textbook, normal. Perhaps I need to try to keep my carb intake at 150 per day. Not too high and not too low. So sick of it all.
I got this article as a pass on from a friend as my 9 yo daughter is experiencing type 1 diabetes issues. We have loved the snack ideas they have been great for helping keep blood sugar levels low. Thank you for this and I look forward to reading more and looking at your recipes too!
Hi, I’ve been looking for this for 6 months. 8 months ago I just have up on GP’s condescending ignorance about reversing T2D. Despite the diet books saying you need medical supervision they’re was none available.
I bought a dexcom g6, as many audio books on the subject as I could get and started walking miles on top of some gym work.
I lost 3 stone in 3 months but the Gp was ignorant of everything and tried to explain A1C. I couldn’t get any further, my meds were halved, weight stayed down despite stopping the fasts, carbs cut to very little.
My body fat according to the gym monitor is about 13%. I need a dexa scan for visceral fat measurement without which I don’t think I can get mich further. [it may be that the term of diabetes and senescence will prevent complete reversal] y I don’t know.
I tried to find private endocrinologists but one wouldn’t answer questions and two weren’t with the reversal movement.
That had to be resolved. But from my point of view, a diet, low in carbs, with regular fasts and plenty of exercise can achieve amazing results.
My wife is going to wear the GGM FOR 10 days and try to eat the same as me. It should be instructive, but yours is the only thing I’ve found.
And I can’t thank you enough for it.
If you want I will send you a short report on our results.
Thank you very much
Seán O’Conghaile
Amazing work! Yes, feel free to share (see the contact page of the site).
Hi there,
I’m a non-diabetic who is considering using CGM to better understand my physiology.
I really appreciated your article. As a researcher myself, it bothers me when people make anecdotal conclusions without any base, so I really like your scientific approach.
However, I am disappointed at not seeing the references at the end of the article. It is a pain to try and find those with only the journal and year, without reference to the authors or a proper reference list at the end.
I understand the average person wouldn’t go looking for a reference list, but in that case it’d be nice if the citations included more information on the papers so that they could be quickly traced.
I’ve looked around GS for a while with Stanford, PLOS Biology, CGM and 2018, but without much luck.
I’ll definitely find it, but I just wanted to let you know :).
It’s impressive how stable your blood glucose levels are!
I’m curious if you’ve tried the effect of any particular supplement or spice (cinnamon, ginger…)?
Many thanks again and have a great day.
Micaela from Switzerland
Found it!
https://journals.plos.org/plosbiology/article?id=10.1371/journal.pbio.2005143
🙂
The references are in fact at the bottom of the post. I realized after reading your comment that you might not have seen the “toggle” button when you click references, so I reformatted them so they are easier for folks to find.
I agree with everything you discovered wearing a CGM.
One of the most interesting aspects of a CGM is that the person can try one food and see how their body reacts to it.
I am a male type 1 using the Freestyle Libre CGM. I switch between a low carb (less than 30 grams carbs) and medium carb (less than 50 grams carbs) only because low carb can get boring. I stay with one or the other (low or medium carb) for months at a time.
Rice spikes my blood sugars worse than ice cream and I can’t eat rice. No, I don’t eat ice cream much but I found this interesting.
Oats also raise my blood sugars a lot, even though oats are a healthy grain. I gave up on eating oats.
I do have 20 fruit trees in my yard (mandarins, peaches, etc) and will eat 1/2 cup of fruit with a meal and this does not raise my blood sugar much. I will eat fruit while exercising and this also will not raise my blood sugars too high. I hope the vitamins and varied diet offset any harm of the fruit sugar. Total fruit eaten is probably less than 10 grams per day.
If I eat less cabs than I end up eating more fat and protein. I asked my endocrinologist if this would be a problem and she said no.
This was a very informative and clearly written article.
Thank you
Lily,
thank you so much for this course article and it’s balanced analysis and recommendations.
I’m a type 1 diabetic, 50years old, healthy, hba1c 6.9.
For years I’ve been curious about low carb but everything send to be written by fanatics. Low carb fanatics. No carb fanatics. Paleo fanatics.
This is a refreshing science based article that re ignites my interest. (we as a family have been giving everyone porridge as a “healthy” breakfast. Now I don’t know what to do (6, 11 and 13 year old kids).
my unanswered concern tensions about the effect of fats and eggs on my own cholesterol and triglyceride profiles
Of you had any helpful links to provide balanced views on that I would greatly appreciate it and I would think many of your audience have the same concern.
Thanks again, from Ireland
Alan
Thanks for this post, I’m currently doing a CGM two week test to check my reactions to various foods, I’m not diabetic.
I had spaghetti bolognese tonight and my levels shot right up – I’m going to have a different evening meal tomorrow to see if the same happens.
Anyway – thanks again.
Great article. I wish there was a date someplace so I could tell how old this info is. Thank you for those pricing guidelines. I have spent a lot of hrs looking for that. Given the cost I don’t think I could afford to do. I need more time than 1 or 2 months. Hopefully the prices will come down. Wonder if it can be purchased cheaper out of the county. I have terrible fatigue swings that I believe in my BS. Been eating keto for 7 years with some success. I still hit the wall if I don’t eat as well as if I do eat. The info I find from people like you who post their results using the CGM has helped me tremendously to understand where I am failing. I LOL when I read the oatmeal and GD, exact what I was told to eat 29 years ago when I was given the GI test. I never was given the actually data from the test. The idea of the CGM is so appealing because it gives me control of the raw data. That is what I want, to be able to see it, study it, experiment. I hope to see more tools like this being placed in consumers hands. Check out the Eyeque tool, great idea for your eyes, needs some help, but on the right path. Thank you again.
Thank you for this amazing article. As a fellow researcher and Google Scholar and PubMed hobby reader I am also currently doing a CGM self experiment. I am generally healthy and have lean-type PCOS and I hoped knowing my blood sugar levels could help me regulate my irregular menstruation. It has been SO eye opening to see your graphs (so different from mine and I thought I eat a healthy low-ish carb diet!) I have many more spikes throughout the day. I was wondering: do you snack in between or do you just eat two big meals a day?
One of the best blog posts I have read in a while! Amazing effort. I couldn’t agree more with everything you have written. As someone who wears a CGM on a regular basis to see how I respond to food – I really do believe it is going to become mainstream for non-diabetics to measure their blood sugar in the next 5-10 years. As a nutritionist, I know no one size fits all – we are all different. Genes, our gut bacteria, stress levels are just a few of the things that impact how well we tolerate carbs. Testing is one of the key ways we can understand which foods we tolerate and which we should avoid for optimal. It was a game changer for me to see foods – we are told are healthy – were sending my blood sugar into pre-diabetic ranges (oatmeal being one!). Brilliant post. Thank you
Glad you enjoyed it, Vanessa. I agree — I think CGM will become more widespread in the coming years and really help clients and practitioners alike make the connection between food and well-being.
This is the best and most succinct account of blood sugar repercussions on health, in general that I’ve read. Your reportage here is exactly what I can send to innumerable friends and family who don’t get what I’m talking about (or don’t care…). And who don’t care to deep-dive into all the implications which I love doing. You can’t read your words above and then look away. And I’ve read everything (only a slight exaggeration) since 2011 when I became obsessed with the implications of sugar/carbohydrates on the health of human beings. (now convinced, incidentally, that the implications on our feline and canine friends are equally compelling)(much gratitude to Gary Taubes and Robert Lustig). Anyway, I love your style and smarts. You are a gift.
Thank you, Mavis!
Thank you Lily for this article. I’m on the other end of the fertility spectrum (Proud Post-ie) but I am very interested in longevity. June 2020 my husband and I were introduced to low carb eating and IF by our family doctor no less. We’ve had much success (relatively pain free), reached our goals, heath and otherwise< and are now fine-tuning maintenance. I'm very interested in wearing a CGM for a while to "see" what actually happens in my body after eating certain foods. I've picked up on some of the subtle physical cues but really need it driven home. I'm not a numbers girl but can read a graph. Reading about your experience lets me see this will be worth the investment. I'm in the US and this would be strictly out-of-pocket as I'm a "norm-ie". Thanks again
You can get a free trial of the Freestyle Libre 14 day sensor by going to the Abbot website and downloading a coupon. You don’t need the reader/kit if you have a smartphone…it works with Apple or Android by downloading the app. I easily got a prescription from my provider. My husband and I both are wearing one, to see who is right about diet and exercise ideology.
Some time ago, I found the same as you found: When my BG was falling fast, I got ravenously hungry. I explain it like this: If your toddler was coming down a slide and there was broken glass at the bottom, you’d try to catch the child *before* they reached the glass rather than waiting until they were cut by the glass. The body is trying something similar: trying to make you eat *before* you go low.
Hi Lily,
I’m currently researching the glucose monitoring system for my wife. Your article was very informative, thank you Just one quick question, did you try adding fat to the oatmeal to see if your sugar levels didn’t spike.
Thank you for your research
Jess
I have just started using a cgm. I am 64, non-diabetic and just want to be in better metabolic health. I am having lots of low readings which are nerve wracking. I wonder if the placement of the probe has anything to do with the lows. I have a very flabby upper arm and mildly overweight. Any advice?
I’ve asked many if a continuous insulin monitor could be possible. We’ll see if the technology gets there (not sure insulin can me measured in interstitial fluid like glucose, I know that’s the challenge with the development of continuous ketone monitors…), but until then the Kraft Insulin Assay is an option. That’s where a glucose tolerance test is given and both glucose and insulin are measured (fasting and post glucose load). See this paper: Crofts, Catherine, et al. “Identifying hyperinsulinaemia in the absence of impaired glucose tolerance: An examination of the Kraft database.” Diabetes research and clinical practice 118 (2016): 50-57.
As to my spike from oatmeal, there’s huge interindividual variability in responses to different foods, even with the same glycemic index or glycemic load. Oatmeal is an outlier for me, while other carbs are less of a problem. All objective measures point to my overall metabolic health. Normal fasting blood sugar, normal average blood sugar, normal A1c, normal fasting insulin, normal HOMA-IR, normal leptin:adiponectin ratio, normal body weight, normal body fat %, normal blood lipids (including advanced lipid profiles looking at particle size and number), normal fertility, and no chronic health conditions.
In subsequent CGM experiments since I published this, I have experimented with eating higher carb for a few days leading up to a high carb, low protein, low fat food, like oatmeal, and my spike is reduced, suggesting my pancreas has temporarily adapted to releasing larger boluses of insulin to cover repeated glucose spikes. This aligns with what we’ve known about glucose tolerance tests since at least the 1960’s and to the animal data. That said, eating that way makes me feel like garbage, so I return to the same place I’ve returned to for over 15 years: sufficient protien, sufficient fat, moderately low carb.
I started using Ambrosia’s NightRider Blucon with Freestyle libre sensor on my endo’s advice and it’s surprising how it gives me the readings on my mobile app without scanning. Moreover, it connects with my Apple watch directly whenever I go out for a run. My life is changed. Highly recommend this CGM combo!
I found this information very interesting. Thanks for doing the research and being a Guinea pig.
Just for entertainment, I got a glucometer. It does kind of suck to stick yourself, but I’m getting better!
I was horrified to find my first fasting BG was 110. I then ate a pastry and a cafe au lait with sugar and an hour later, it was 107. Next morning, fasting was 99. Did not eat anything and did a light workout. 101. Ate chocolate covered “protein bar” (ie candy bar with whey or whatever) and was at 104 two hours later.
I was curious because folks I work with who are health enthusiasts (wear the whoop) and are anti-carb (bulletproof etc) are always wondering how I can be 55, eat cookies and pasta most days but still weigh less than 110 lbs without really following an exercise program.
I think I am opposite you – run high normal values, but have low variability. Then again, I’ve only been checking a few times a day for a few days. Maybe I need to spring for a fancier machine than the $9 fingerstick one at Walmart.
Anyway, I don’t seem to get sugar highs or lows either clinically or on the glucometer. No DM in my family although some obesity. Htn with strokes in the 80s but never MI/CAD.
Data is fascinating. Wish there was more. I’m sure there will be.
Awesome article. Just what I was looking for. I’ve commited to wearing a Dexcom 6 CGM for 90 days, and look forward to analyzing my data.
I just mow found your article. I’ve been crossing the line between pre-diabetes and Type-2. I’m 58 and don’t want to go down the diabetes road and all it’s complications. I recently fasted for 5 days (but ingested coconut oil and avocado oil) in order to clear out the glycogen in my liver and muscles. I am currently following the Atkins 20 diet. I was wondering about how low is too low for blood glucose levels. I’m below 70 a lot and occasionally gone below 60, but by body adjusts quickly. I never have symptoms of hypoglycemia.
I can’t seem to find the information I’m looking for about if I’m running on ketones, my blood glucose SHOULD be low. But is there a “too low” or true hypoglycemia in this situation?
Also, have you repeated your oatmeal experiment with fat and protein added?
Yes, the spike is lessened somewhat, but it’s still not great for my blood sugar compared to a breakfast like eggs, sauteed veggies sourdough, and butter.
Look up zoe. They have you at standardized muffins and check sugars and postprandial lipid levels. Then you wear the cgm for rhe remainder of the 2 weeks and do suggested food experiments. In the end they send you a report with tailored dietary advice. Im in the first week now so cant comment any further but i love the concept
I am also in the first week on Zoe and finding the blood sugar reading very interesting and informative. I can already associate a tired feeling with higher blood sugar readings. Also when my blood sugar readings are higher it affects my sleep. Really interesting stuff! I also believe that we are individuals and will react different to different foods based on our age, microbiome and previous diet preferences.
I am very similar to you as a oatmeal leaves me hangry shortly afterwards. White rice will have me asleep in no time. I was borderline gestational diabetic with both of my kids. As a woman in my early 50’s I am interested in how the hormone changes during this phase will affect insulin and glucose levels . As a nutrition health coach and personal trainer I live continuing to learn about glucose monitoring to help myself and my clients.
This is a pretty fascinating art project
Hi Lily!
I was wondering if you could do the date experiment with the CGM after a whole days fast? It is currently the month of Ramadan, and people are fasting (no food / no water) until sunset (12+ hours). To break your fast, it is recommended to start with a date and then move on to eating “real food”. I saw in your other article that you mentioned you should have dates after food like a dessert. It would be an interesting experiment to see why it is recommended to start with a date after a whole days fast. Thanks!
Lily,
What a great read! Thank you. Blood glucose regulation is so important (as I’m learning).
In any of your research, were you able to find consensus on what an optimal glucose variability would be? Say a range of 40 mg/dL or fewer? What would ‘keeping it tight’ look like?
On a daily basis I found my reading going between 55-155 occasionally, between 70-130 often, ~30% of my day spent under 70 (with symptoms), and symptoms when my blood sugar would spike and the dip rapidly.
Headed to my doctor soon to see if we can’t rule out pancreas issues, and then see if I can’t get a longer test (signed up for one 14-day trial with a Zoe gut biome test I bought) and try to identify the very best foods and food pairings for me that will keep me in my best range all day.
TIA
i was diagnosed with prediabetic i found it interesting how oatmeal raised your blood sugar level. if been picking my finger and found outmeal lowered my blood suger level. Are you sure it wasn’t the honey. I dont like oatmeal im just experimenting. I really do not like drugs so I’m tring to get out of this prediabetic situation with food. Im 64 years old the more i read the more confused I get. This article has helped me understand a lot more. Thank you for wearing the monitor.
Both honey and oats raise blood sugar. You can see a follow up to this experiment here, where oatmeal was tested without honey and with added protein/fat.
One possibility for your findings is that if you only test at 1 or 2 hours after eating, you can miss the spike. If you have a CGM, you’d see the full picture.
Wow! I “stumbled” across this article while searching for a non-diabetes book! A1C and blood work look great! I feel like crap!?? NO ONE WILL LISTEN TO ME! I shake, I sweat, I get mad, i am always hungry, can’t sleep get nauseous i constantly have to eat. Thank God, I found this article. I am going to try the low carb diet and see if this helps. Thank you for caring enough to care!
Thank you so much for this- I’m using a CGM for 6 months to understand how I need to eat in perimenopause with DM2 and high cholesterol in all of my immediate family. Your artice helped me to understand my data a little better!
Something cool that you missed is you actually don’t need the reader! You can just scan the sensor with your phone and the libre app and it tells you your sugars!
On the new Libres, yes this is the case. When I did this experiment and wrote this article, the only available option was to scan with a reader. It’s easier now not having to use a separate device!
Thanks for all your research and putting this all together! For someone struggling to control prediabetes, I find your “naked carb” research relateable to my situation. I can’t wait to put this to work and check my glucose average in 2 weeks.
This is a really helpful article. I was having really weird fasting glucose tests that didn’t seem to match my A1C and this occurred repeatedly year after year, so finally, I asked my doctor for a freestyle libre script and told him I was fine to cash pay, as I knew my insurance wouldn’t. I just wanted to learn more. He was super down for that.
It’s been quite the experiment, and tough because there isn’t a lot of data out there for non-diabetics. My husband has done it now too.
Now the doctor isn’t gonna do a lot of looking at it aside from answering specific questions I bring to him, because monitoring it has costs, but I still thought it was worth doing.
I came across this article while searching for clinical data on blood glucose variability throughout the day in non-diabetic adults.
My interest in this topic was stimulated by a new smart watch that measures serum glucose every 5 minutes via PPG (i.e., similar to a CGM, albeit noninvasively), which I recently purchased.
After calibrating the smart watch with actual fingerstick glucose readings, I was very intrigued by the postprandial glucose values, and particularly by a spike (130s-145) after 12-14 hours of fasting (I do IF for 16-18 hours or day). This daily fasting spike, doubtless brought on by the onset of glycogenolysis, followed by gluconeogenesis, occurs consistently between 0900 and 1200, 7 days a week.
Like you, I found very little published high-level clinical data in this area.
Thanks for a very interesting and well-written / well-researched article!
Robert A. Wascher, MD
Very interesting read. I have PCOS, not diabetic but currently wearing a CGM. It was not easy to find information about what how a “normal” blood sugar fluctuates. I have to admit, it is kinda fun seeing how I react to different foods. I 100% agree, naked carbs kill and rice makes my sugar sky high. I think everyone should have a chance to study their own bodies.
An excellent article!!!! As a guy I really benefitted as u Mom’s got a lot going on metabolically to us mere men.
My search was what is an ideal glucose pattern for a day. I got a cgm and initially this just helped identify spikes. Some dietary and some stress. I never dreamed fear could spike glucose more than carbs. Cortisol, adrenaline, ect,,, ( I have scary job – I love)
Now after resolving my T2D, to normal A1c’s to 4.9 – 5..6, I wanted to insure I did not slip back to T2D.
Just doing a no/low carb diet (<20g) did seem to be enough.
Although this article focuses on glucose I have found my LDL and BP have also migrated towards the ideal range with my change in glucose levels.
So if I can optimize my glucose I hope to optimize my LDL & BP.. This my search for ideal glucose pattern.
I want yours ! Mine is flat now and stress can drive to 160+, But food seldom over 130 now. My bottom is 78, so lines pretty flat now. Which I now know from this article could be flatter but is pretty good, and I’m on statins so once happy with glucose I will adjust that aspect of my health.
Thanks for a really educational article.
Hey Lily,
First, thank you so much for the information you put out there and for Real Food for Pregnancy! I admittedly have a sweet tooth, so my focus has been to incorporate more real food without trying to remove some of my treats, but this has worked to push some of those extra carbs and sugars out of my diet naturally. I asked my doctor if I could use a CGM to test for GD this time and she was fine with it and wrote me a script. I’m only a few days in and so interested. I’m going to be experimenting a lot over the next few weeks with my diet to see what my numbers do. So far I’ve seen a slightly high fasting number (low 90s) but my daily range is 90-115 with a few spikes into the 120-130s after a carb heavy snack. Anyway, thanks so much for this article and all your other research. It’s appreciated (and yikes, epigenetics, sorry to my oldest)!
I am a non-diabetic that wore a CGM for 3 months. I found your article very interesting and what agree with a lot of your findings. I think the one thing that is missing is also the effect of cortisol and adrenaline on blood sugar levels. The same food could give a different result based on the stress level that day that the individual is experiencing. The correlation I was able to make is the higher the cortisol level, no more dramatic effect a food would have.
Yes, stress absolutely affects blood sugar levels, more so in some individuals than others. CGM can help identify this effect as well!
I could not agree more; “huge opportunity in preventative medicine”. Am currently wearing a CGM (Dexcom G7) for the first time as a means of self-study. Of course I self-paid for the sensors & had to buy a new phone to use the app =)~ so I’m trying not to make the most of this opportunity & my investment. I am hoping the first 24 hours are simply a further “warm up” for the sensor, as it’s been quite erratic. I am closely following all instructions, yet my glucose is jumping from “urgent low” of 48 to 118 in less than 5 minutes. trying to learn if this is normal? It’s happened a few times, not sure why, but I’m working on it…& trying not to fret.=)
I have no diagnosis per any lab work, though my A1C tested at 5.7 a couple of years back, which is technically pre-diabetic, as I understand it. Doctor suggested I start taking a red rice yeast supplement, which I bought but did not take. I have been eating a meat-based diet for a few years now, less than 20 carbs a day overall, & less than 10 most days. I’ve also been using a Keto Mojo meter off & on during this time, & am at a healthy weight.
I love that this technology is available, & firmly believe it can go a long way toward avoiding any further diagnoses, as well as gathering info on daily habits.; exercise, food, sleep, stress, etc. Your article is the first to pop up when I searched for “typical CGM graph non-diabetic, self-study”, as an aside~ just FYI.
Thank you so much for sharing your data & thoughts ~ the more I learn, the more I want to learn. At 58 years old, I am post-meno & committed to optimizing my health and longevity. So grateful for the tools, education, and experience of others to add to my own.
This is exactly the information I was looking for. I am non-diabetic and have a CGM and respond very similar to what you describe. I also have sleep apnea and I think that is making my A1c in the prediabetic range. Doctors keep wanting to treat me with metformin and I say I need the sleep apnea treated. My fasting insulin, HOMA-IR, fasting glucose and lipid ratio are really optimal. CGM shows good insulin sensitivity. Thanks for the comprehensive article that helps me make informed decisions.
Diabetes has a large genetic component. I read that if 4 people with identical twins have it, 3 of their twins do too.
“ The point is that even if you’re “metabolically healthy,” your blood sugar can still spike pretty high if you eat a bunch of naked carbs.”
Sums it up beautifully. Spikes after eating carbs are normal and don’t mean you have Type 2 Diabetes. But if you do that every meal for 50 years, you may end up with it!
Dear Lily,
I recently read your insightful blog post about your experience wearing a continuous glucose monitor (CGM) as a non-diabetic, and it prompted me to reflect on some interconnected aspects of blood sugar regulation and cardiovascular health.
When we consume foods high in sugar or refined carbohydrates, our blood glucose levels can spike dramatically—a “rollercoaster” effect you’ve described. The body has a few primary mechanisms to manage these surges:
Glycogen Storage: Excess glucose is stored as glycogen in the liver and muscles, but this capacity is limited.
Conversion to Fat: Once glycogen stores are full, additional glucose is converted into fatty acids through a process called de novo lipogenesis.
VLDL Production: These newly formed fats are packaged into very-low-density lipoproteins (VLDL) by the liver and released into the bloodstream. VLDL can eventually become low-density lipoproteins (LDL), which are associated with an increased risk of coronary artery disease, stroke, and other cardiovascular issues.
From an energy perspective, considering that our bloodstream only contains about a teaspoon of sugar at any given time, ingesting large amounts of sugar requires the body to act swiftly to restore balance. When storage options are maxed out, converting excess glucose into fats becomes a necessity, leading to higher levels of circulating lipids.
This brings me to a concern: It seems plausible that a significant portion of the population—perhaps 80% of Americans consuming high-sugar diets—may have elevated levels of fats in their blood due to this metabolic response. Yet, the link between high sugar intake, VLDL/LDL production, and cardiovascular disease doesn’t seem to be prominently addressed in public health discussions.
My questions are:
Why do you think this critical connection is often overlooked or not made clear to the public?
Or is there an effort to downplay this connection by blaming LDL levels to ingestion of saturated fats.
In the context of your CGM findings and nutritional expertise, do you see this as an area that needs more attention?
Did I miss any key aspects in my understanding of how excess sugar intake leads to increased VLDL and LDL levels, and consequently, cardiovascular risks?
I believe that highlighting this link could be vital for better public awareness and preventive health measures. I’d greatly appreciate your thoughts on this matter.
Warm regards,
Rex
I’m non-diabetic as well but decided to try the monitor for 14-days. I’m at the end of my “trial” and I’ve noticed, and I haven’t changed much in my habits, but as the hour is approaching where my device “expires”, seems my glucose readings are up by a whole 1-point. Could that be the company programmed the device to do this so to sway me to get another before time is up? Perhaps another experiment is due.
Likely not, is just a “dying” chip. I’m also non diabetic and try to understand my body which has some weird relationship with food.
Once it happened in the last 3 hours that the readings went up and down with sometimes even 10 points within a minute…. that can happen with technology. You are usually not going to buy a new sensor for such thing. It is mostly used by diabetics who will buy another one anyway.
Loved this article – can you tell me when it was written? (I find it useful to have dates for when things are written or updated as I make notes in my journal on what I’m studying.) I’m currently using the new Stelo (as of Sep 2024) and am finding it fascinating during my low carb journey. Thank you so much for citing your research at the end of the article!
2018!
Is it normal for my child to have a high fasting blood sugar reading of around 120 each morning? Age is 8. I have prediabetes and since he has a lot of my same symptoms I decided a few finger pokes couldn’t hurt…
I really appreciated your CGM experiment post, Lily; it was both insightful and grounded. Your emphasis on not overreacting to every glucose fluctuation and focusing on the bigger picture can be applied to most things in life.
This was a balanced perspective in a space that often leans toward data obsession.
Given your interest in understanding glucose patterns without being floaded in constant notifications, I wanted to share something: someone at the TRMNL community developed two plugins that allow Dexcom and Nightscout data to be displayed in our minimalist e-ink screen.
It’s a subtle way to keep an eye on glucose trends without the distractions of apps or alarms.
If this piques your interest, we’d be delighted to send you a complimentary device—no strings attached. We’d genuinely value any feedback or thoughts you might have.
Thank you for sharing your experience with a continuous glucose monitor! Reading about how your glucose levels changed throughout the day really opened my eyes to how our bodies react to food and activity. It makes me want to try it myself to understand my health better.
Absolutely brilliant article and insightful research! I have my own conspiracy theory. The official nutrition guidelines are chosen this way because if they told the people the real truth, which is that carbs aren’t really healthy, it would be impossible to feed all these billions of people on earth. Rice and wheat harvests are trillion dollars per year. I believe it would be absolutely impossible to feed all humans with appropriate amounts of animal or fish protein and sufficient healthy fats.
It’s a bit of a “Soylent green” realization for me. The cgm proved to me beyond doubt that eating carbs, even the so-called healthy ones is definitely not a good idea. Unless you eat them with tons of fat, which is another point where reality and nutrient politics diverge in opposite directions….
March 2026 update: Stelo by Dexcom is available without prescription. Order online. $55 per sensor lasting 15 days. Uses Bluetooth to transmit data to phone app. No separate reader. I’m on week 3. Process is easy & painless. I’m not diabetic but was curious about how foods affect energy levels.